This unit will include an overview of the main sources of indoor air pollution (breathing and carbon dioxide emissions in highly occupied classrooms, cigarette smoking, use of electronic cigarettes and emissions of organic compounds resulted from the use of cleaning agents, building materials, etc.). Students will learn about the differences between inorganic and organic compounds, aromatic hydrocarbons, perform experiments and do computations to solve real life problems involving moles, concentration, volume and rates of change for different chemicals. They will use carbon dioxide monitors to compare and contrast the carbon dioxide concentrations inside and outside the classroom, as well as studying what happens when a classroom is ventilated properly.
1. Sources of Indoor Air Pollution
Part 1 is a review of indoor air pollutants. Indoor pollution sources that release gases or particles into the air are a primary cause of indoor air quality problems. Inadequate ventilation can increase indoor pollutant levels by not bringing in enough fresh outdoor air to dilute emissions from indoor sources. High temperature and humidity levels can also increase concentrations of some pollutants through the higher release rate of some chemicals with temperature or the growth of mold on damp building materials. The following source descriptions provide emphasis on the air pollutants (asbestos, carbon monoxide, formaldehyde and pressed wood products, lead, nitrogen dioxide, radon, indoor particulate matter and sources such as secondhand smoke/tobacco smoke, stoves, heaters, fireplaces and volatile organic compounds) [8].
Asbestos
is a mineral fiber that occurs in rock and soil. Because of its fiber strength and heat resistance it has been used in a variety of building construction materials for insulation and as a fire-retardant. Asbestos has been used in a wide range of building materials, such as roofing shingles, ceiling and floor tiles, paper products or asbestos based cement products.
Elevated concentrations of airborne asbestos can occur after asbestos-containing materials are disturbed by cutting, sanding or other remodeling activities. Improper attempts to remove these materials can release asbestos fibers into the air in buildings, increasing asbestos levels and endangering people living in those homes.
Exposure to asbestos increases the risk of developing lung disease including cancer. That risk is made worse by smoking. In general, the greater the exposure to asbestos, the greater is the chance of developing harmful health effects.
Carbon monoxide (CO)
is an odorless, colorless and toxic gas. Because it is impossible to see, taste or smell, CO can kill humans before they are aware it is in their home. The effects of CO exposure can vary greatly from person to person depending on age, overall health and the concentration and length of exposure.
Carbon monoxide is produced during the incomplete combustion of wood and fossil fuels. Sources of CO include: unvented kerosene and gas space heaters, leaking chimneys and furnaces, back-drafting from furnaces, gas water heaters, wood and gas stoves, fireplaces, generators and other gasoline powered equipment, automobile exhaust from attached garages and tobacco smoke
Exposure to carbon monoxide leads to the formation of carboxyhemoglobin in the blood, which inhibits oxygen intake. At low concentrations, the health effects are fatigue and chest pain; at higher concentrations, impaired vision and coordination, headaches, dizziness, confusion and nausea. CO exposure is fatal at very high concentrations.
Formaldehyde (HCHO)
is a colorless, flammable gas at room temperature and has a strong odor. It is an important chemical used widely by industry to manufacture building materials and numerous household products. Also, formaldehyde is a by-product of combustion and other natural processes. Thus, it may be present in substantial concentrations both indoors and outdoors. Formaldehyde can cause irritation of the skin, eyes, nose, and throat. High levels of exposure may cause some types of cancers [12].
Lead (Pb
) has long been recognized as a harmful environmental pollutant. Lead is particularly dangerous to children because their growing bodies absorb more lead than adults do and their brains and nervous systems are more sensitive to the damaging effects of lead. Babies and young children can also be more exposed to lead because they often put their hands and other objects that can have lead from dust or soil on them into their mouths. Children may also be exposed to lead by eating and drinking food or water containing lead or from dishes or glasses that contain lead, inhaling lead dust from lead-based paint or lead-contaminated soil or from playing with toys with lead paint. Before the health effects and environmental distribution of lead were fully understood it was used in paint, gasoline, water pipes, and many other household products.
Nitrogen dioxide (NO
2
)
The two most prevalent oxides of nitrogen are nitrogen dioxide (NO
2
). NO
2
is a highly reactive oxidant and is corrosive. The primary sources indoors are combustion processes, such as unvented combustion appliances (gas stoves), appliances with defective installations, tobacco smoke and kerosene heaters. Nitrogen dioxide acts mainly as an irritant affecting the eyes, nose, throat and respiratory tract [13].
Extremely high-dose exposure (as in a building fire) to NO
2
may result in pulmonary edema and diffuse lung injury [13]. Continued exposure to high NO
2
levels can contribute to the development of acute or chronic bronchitis. Low level NO
2
exposure may increase the risk of respiratory infections, especially in young children. Average level in homes without combustion appliances is about half that of outdoors. In homes with gas stoves, kerosene heaters or un-vented gas space heaters, indoor levels often exceed outdoor levels.
Radon (Rn
) is a naturally occurring radioactive gas that can cause lung cancer. Is seeps into buildings from the surrounding soil and rocks. In some cases, well water may be a source of radon. The U.S. EPA ranks indoor radon among the most serious environmental health problems facing humans today. After smoking, it is the second leading cause of lung cancer in the United States causing an estimated 21,000 lung cancer deaths a year.
A nationwide survey of radon levels in schools estimates that nearly one in five has at least one schoolroom with a short-term radon level above the action level of 4 pCi/L (picoCuries per liter) - the level at which U.S. EPA recommends that schools take action to reduce the level. U.S. EPA estimates that more than 70,000 schoolrooms in use today have high short-term radon levels [9].
Indoor particulate matter
(also referred to as PM or particle pollution) is a complex mixture of solid and/or liquid particles suspended in air. These particles can vary in size, shape and composition. The U.S. EPA is especially concerned about particles that are 10 micrometers in aerodynamic diameter or smaller (PM
10
) because these particles are inhalable and too small to rapidly settle out of indoor air. Once inhaled, particles can affect the heart and lungs and in some cases cause serious health effects.
Indoor particulate matter can be generated through cooking, combustion activities (including burning of candles, use of fireplaces, use of unvented space heaters or kerosene heaters, cigarette smoking) and the re-suspension of floor dust due to indoor high occupant activity. A recent study indicate that 70% of indoor fungal aerosol particles and 80% of airborne allergenic fungi were associated with indoor emissions. On average, 81% of allergenic fungi from indoor sources come from occupant-generated emissions [5].
2. Is CO
2
an Indoor Pollutant?
Part 2 will address carbon dioxide in buildings. Because humans produce and exhale carbon dioxide (CO
2
), concentrations in occupied indoor spaces are higher than concentrations outdoors. As the ventilation rate (i.e., rate of outdoor air supply to the indoors) per person decreases, the magnitude of the indoor-outdoor difference in CO
2
concentration increases. Consequently, peak indoor CO
2
concentrations, or the peak elevations of the indoor concentrations above those in outdoor air, have often been used as rough indicators for outdoor-air ventilation rate per occupant. The need to reduce energy consumption provides an incentive for low rates of ventilation, leading to higher indoor CO2 concentrations [3].
The graph below (Figure 1) shows that in the absence of ventilation, CO
2
concentrations in a room with closed windows and doors could increase to 1000 ppm within 45 minutes of enclosure.
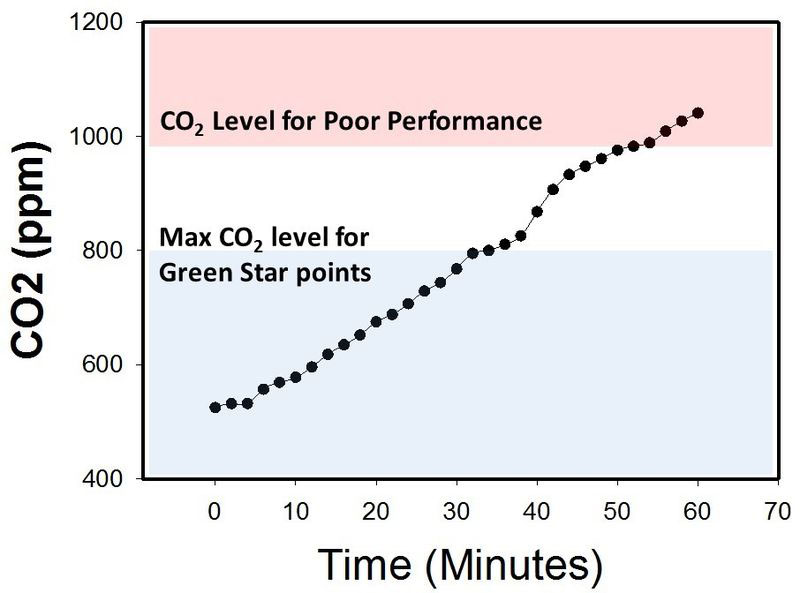
Figure 1.
Carbon Dioxide concentration in a closed room with no ventilation.
Source: https://en.wikipedia.org/wiki/File:Edaphic_Scientific_CO2_Levels_for_HVAC_and_IAQ.jpg
Concentrations of CO
2
inside highly occupied buildings range from outdoor levels up to more than 3,000 ppm. Prior research in industrial settings has documented direct health effects of CO
2
on humans, but only at concentrations much higher than those found in normal classrooms indoor settings. CO
2
is the key regulator of respiration and arousal of behavioral states in humans. The initial effects of inhaling CO
2
at higher concentrations are increased partial pressure of CO
2
in arterial blood and decreased blood pH [3]. CO
2
concentrations greater than 20,000 ppm cause deepened breathing; 40,000 ppm increases respiration markedly; 100,000 ppm causes visual disturbances and tremors and has been associated with loss of consciousness; and 250,000 ppm CO
2
(a 25% concentration) can cause death. Maximum recommended occupational exposure limits for an 8-hr workday are 5,000 ppm as a time-weighted average, for the Occupational Safety and Health Administration (OSHA 2012) and the American Conference of Government Industrial Hygienists.
A recent study conducted at the Lawrence Berkeley National Laboratory (LBNL) has challenged the conventional wisdom that no health effects result from the CO
2
concentrations typically found in occupied settings such as school classrooms [3]. Twenty-two participants were exposed to CO
2
at 600, 1,000, and 2,500 ppm in an office-like chamber. Each group was exposed to these conditions in three 2.5-hr sessions, all on 1 day, with exposure order balanced across groups. At 600 ppm, CO
2
came from outdoor air and participants’ respiration. Higher concentrations were achieved by injecting ultrapure CO
2
. Ventilation rate and temperature were constant. Under each condition, participants completed a computer-based test of decision-making performance as well as questionnaires on health symptoms and perceived air quality. Participants and the person administering the decision-making test were blinded to CO
2
level. Data were analyzed with analysis of variance models and the results are presented in Figure 2.
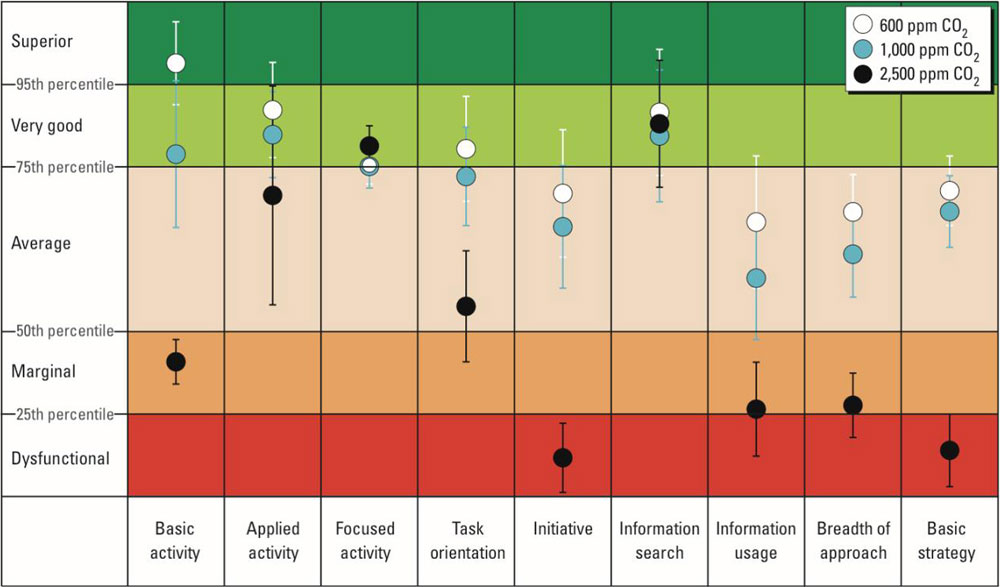
Figure 2. Impact of Carbon Dioxide on human decision-making performance.
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3548274/
The results were as follows: relative to 600 ppm, at 1,000 ppm CO
2
, moderate and statistically significant decrements occurred in six of nine scales of decision-making performance. At 2,500 ppm, large and statistically significant reductions occurred in seven scales of decision-making performance (raw score ratios, 0.06–0.56), but performance on the focused activity scale increased.
The conclusions of the study was that direct adverse effects of CO
2
on human performance may be economically important and may limit energy-saving reductions in outdoor air ventilation per person in buildings [3].