Foundation: Coulomb’s Law
The foundation of this unit is Coulomb’s law, which describes the relationship between electrically charged particles. At the level of the atom, this force exists between negatively charged electrons and positively charged protons, which both have a magnitude of one. As the phrase goes, “opposites attract,” and this rings true not only in love but in chemistry as well. Protons and electrons are attracted to one another. And just like in love, distance can put a strain on this attraction. Coulomb’s law states that the size of the attraction depends on not only the size of the positive and negative charges but on the distance between charged particles as well. Imagine there’s a love interest sitting next to you in chemistry class in the morning and at the end of the day, you’re in separate classes one floor away. In which scenario will your interest and attraction be higher? Most would agree that sitting so close to a love interest quickens the heart and affections much more than being out of sight of one another. Similarly, the attractive force generated between a proton and electron one angstrom away is much stronger than the force between the same two particles 3 angstrom away. The closer the charges are to one another, the stronger the attraction.
On the other hand, Coulomb’s law also explains that similar charges repel one another. It is not uncommon to see a couple or friends or family members that are so similar that they just cannot get along. In chemistry, like charges repel each other and generate a force of repulsion that is also affected by the size of the charges and the distance between these charges. Electrons that are too close will generate a strong repulsion, as do protons that are too close.
This simple law extends well beyond just protons and electrons. Many other things in chemistry can be charged as well, including an entire atom that has lost or gained an electron becoming an ion or a collection of atoms known as a polyatomic ion or even macromolecules like proteins and DNA. Unlike protons and electrons, which only have a charge of one, ions can carry a one, two, three, and sometimes even four negative or positive charge. Polar molecules are molecules having a section with a partial charge known as a polar region by the unequal sharing of electrons between atoms. In these cases, the size of the positive or negative charge is much more variable. The partial charge on regions of molecules are less concrete but can be generally measured by looking at the electronegativity values of atoms sharing bonding electrons.
Teaching strategy: “You’re under arrest!” Coulombic Attraction
Coulombic attraction is a foundational concept that is critical for students to understand for this unit. After briefly introducing the information, randomly assign half of your students to be a positive particle, negative particle, or neutral using red shirts for positive, blue shirts for negative, and white for neutral. While half of your class watches, have your participating students enter the room into a marked out area one by one and begin interacting with one another using the principle of Coulombic attraction. Participating students should be constantly moving (just like all particles do) and should be either moving away from other identical particles or moving towards opposite particles. To avoid students of opposite charges remaining static next to each other, encourage students to keep moving even if it’s together. Eventually the pair should separate when they encounter other oppositely charged particles. Students who are watching can be Coulombic officers looking for violations of Coulombic attraction. When violations are observed, officers can “buzz” halting all movement while they point out the violation and “arrest” the suspects. These violators can swap out with the officer giving students a chance to play either role.
Electronegativity and bonding
Electronegativity is the measure of how strongly an atom will pull shared electrons in a bond towards itself; electronegativity values range from 0.0 to 4.0. If two atoms sharing electrons in a bond have a difference of electronegativity greater than 2.0, electrons will be transferred to the atom with larger electronegativity and the two atoms will become ions with opposite charges. Coulombic attraction will attract these two ions together, creating an ionic bond. If the two atoms differ in electronegativity between 0.4 and 2.0, the electrons will be pulled closer to the atom with larger electronegativity and the bond is considered a polar covalent bond. A difference in electronegativity less than 0.4 is considered a nonpolar covalent bond and electrons are shared equally in the bond.
As long as there is some kind of negative and positive charge in a molecule, there will be some kind of repulsion and/or attraction. The attraction between the negative electrons and positively charged nucleus causes electrons to order themselves as close to the nucleus as possible, while the repulsion they experience due to neighboring electrons forces electrons to maintain their distance from one another. These simple phenomena of attraction and repulsion continue at a larger and larger scale determining the geometry of larger and larger molecules.
Teaching strategy: Types of Bonding based on Electronegativity
In this activity, students will practice determining the type of bond made between atoms based on electronegativity differences. Make large index cards with an element from the first 3 of 4 rows written in letters large enough to see from the back of the room and the electronegativity value written on the other side. Make multiples of common elements like H’s, O’s, C’s, and N’s. Give each student a periodic table with electronegativity values listed as well. Call students up randomly to the front to draw an atom and make a bond. Have these two student display their atoms to the class and race to figure out if the bond is polar covalent, nonpolar covalent, or ionic. If you choose to make the competition only between the two students standing, the winner can stay to race the next student or two new students can be called up to participate. Alternately, you could create a bracket with pairs decided ahead of time to race; winners go on to compete against each other and losers compete against each other in the losers' bracket. The competition can also be for the entire class and students could write down answers and check for the most correct at the end.
Stability from Bonding
Finally, in this foundational section, students will learn that atoms form bonds to create more stability by lowering the energy of each bonding atom. Just like teenagers (and adults), molecules in chemistry choose the option that requires the least amount of energy. Repulsion between like particles causes the energy of that particle to increase sharply while the attraction between oppositely charged particles causes the energy of the particles to decrease. Because protons and electrons exist within atoms that contain
both
protons and electrons, finding the “sweet spot” of high attraction and minimal repulsion can be difficult. Attraction and repulsion between charged particles are automatic and cannot be helped; imagine the moth that just can’t escape the allure of the light bulb. Atoms are attracted to one another because the outer negative electrons of one atom are attracted to the positive core of protons in nucleus of the other atom. However, this attraction is quickly blocked by the repulsion between the outside electrons of both atoms. Getting too close takes too much energy but being too far away does not lower the energy and promote stability of both atoms; somewhere in the middle is just right.
Connecting Structure and Properties
Why is butter a solid at room temperature while oil is a liquid? Why does acetone evaporate so easily when spilled while water can sit for much longer? Why can I dissolve so much more salt in water than sugar?
Every type of substance has its own unique properties, which can be partially explained using its chemical structure. This connection is captured in the central mantra of biochemistry: “structure determines function.” This mantra can be seen all around us from the equipment we use to the particular body builds that are best suited for sports like gymnastics, sprinting, or swimming. In the microscopic world of molecules, this mantra still rings true. The function and properties of molecules arise from their shape and bonding between atoms.
Perhaps the easiest property to explain about a substance is why it is a solid, liquid, or a gas at room temperature. We know that butter and oil are very similar substances and can often be substituted for one another in baking recipes. However, oil is a liquid at room temperature while butter is a solid. Why is this?
Phases of matter differ in how closely packed the individual molecules of that substance are to one another. Let’s use water as our example as we daily see water existing at all three phases of matter. When water is solid ice, its individual water molecules are very packed together, slowly vibrating in place. On the other hand, when water is heated past 100
o
C, it turns into steam, the gaseous form of water and its molecules are moving incredibly fast and are spread very far apart from one another. In the normal liquid phase, water molecules are loosely spaced and moving at a medium speed compared to the gas and solid forms of water. For molecules of any substance to exist as liquids and solids at room temperature, there must be some kind of attraction between molecules keeping them more closely packed together. As stated earlier in the foundation of this unit, the only type of attraction that exists between molecules is Coulombic attraction between positive and negative charges. Consequently, substances that exist as solids at room temperature must have many negative and positive charges/regions and have the strongest attraction between their individual molecules and substances that exist as gases have very weak attractions between individual molecules that can be easily broken and consequently have a lack of positive and negative regions. Since all molecules have protons and electrons, what makes one substance’s molecules much more positive and negative than another’s?
The answer lies in how the atoms are arranged in the molecule and how equally or unequally electrons are distributed, something determined by the electronegativities of each atom. If two atoms are connected in a molecule that have very different electronegativities, like hydrogen and oxygen, electrons will not be shared equally but instead will stay closer to the atom with greater electronegativity. This lopsided distribution of electrons will cause one atom to become partially negative and the other to become partially positive. And because a substance contains millions of these molecules, all with unequally distributed electrons and partial negative and positive charges, these molecules will be attracted to one another, arranging themselves to allow the positive side of one molecule to be close to the negative side of the next molecule.
As the polarity in a molecule increases, the ability of the individual molecules to stay attracted and close to one another also increases, ensuring that the substance will remain a solid at higher and higher temperatures. Remember that higher temperatures means each molecule is moving with greater kinetic energy, putting an increasing amount of strain on the intermolecular attraction keeping molecules together. Consequently, if a substance has a high melting and boiling point, this substance must have very strong attractions between its individual molecules. In the next section, specific types of attractions between molecules will be explained.
Teaching strategy: “Am I a solid, liquid, or gas?”
To better understand how a substance’s phase of matter depends on attraction between molecules, create multiple molecular models of 3-5 different substances that are solid, liquid, or gas at room temperature. Each substance should have its own station around the room containing a box of 5-10 models of that molecule. In groups, students visit a station and arrange the molecules obeying Coulombic attraction. The greater the points of attraction, the closer together molecules will be, illustrating a substance in solid state. Water and hydrochloric acid could be used for liquid, oxygen and methane for gas, and glucose for solid. It’s important to note that phases of matter are dependent on far more than just the number of attractions between molecules of that substance. For example, sulfur hexafluoride has 6 polar bonds but because of its symmetry and shape, this polarity cancels out and this substance is a gas at room temperature. Also, very large molecules like hydrocarbons tend to be solids even without polar bonds due to the large amount of van der Waals forces between molecules.
Intermolecular Forces
In the third part of this unit, students will then learn specific kinds of forces called intermolecular forces (IMF) between molecules including London dispersion forces, hydrogen bonding, and hydrophilic and hydrophobic attractions.
The first type of attraction between molecules, dipole-dipole interactions, is in substances whose molecules have permanent polar bonds in which electrons are unequally shared between atoms. This uneven distribution of electrons leads to a permanent positive and negative regions of the molecule leading to a strong attraction between molecules of the substance. The positive region of one molecule will be attracted to the negative region of a neighboring molecule like bar magnets, only with charge instead of magnetism.
A special type of dipole-dipole interaction between hydrogen atoms and the most electronegative atoms (fluorine, oxygen, and nitrogen; chlorine is not included because of its larger size) is also identified known as hydrogen bonding. The electronegativity difference between these atoms is so large that these dipole-dipole interactions are particularly strong and account for especially high melting and boiling points that deviate from periodic trends.
These first two types of attractions do not, however, account for how large molecules without polar bonds are able to exist as solids like butter and wax. In these cases, an attraction called London dispersion forces plays the prominent role in keeping these otherwise uninterested molecules together. London dispersion forces explain that molecules can experience temporary dipole moments where electrons are momentarily lopsided on one side of the molecule creating a partial negative and positive region in the molecule. This temporary dipole moment in a molecule creates a domino effect on the molecules around it, causing electrons to shift in each molecule to whichever region of the molecule will reduce the repulsion to the negative partial charge in neighboring molecules and increase the attraction to the positive partial charge. These temporary dipole moments in the collection of molecules lead to attractions between molecules, keeping them together. Though this IMF is relatively weak, it plays a more prominent role in large molecules or in molecules made of atoms with high atomic numbers that have a greater number of electrons, thus increasing the likelihood of temporary dipole moments.
It should be noted that there is a distinction that should be emphasized between attractive forces
within
the molecule
between atoms
called intramolecular forces and the forces
between molecules
called intermolecular that was discussed above. Intramolecular forces lead to covalent or ionic bonding while intermolecular forces lead to properties like melting point and boiling point.
The final type of interaction that will be discussed is hydrophilic, “water-loving” and hydrophobic, “water-hating” interactions. Water is a small molecule made of two hydrogen atoms bonded to one oxygen atom. As explained earlier, the hydrogen-oxygen bond is an extremely polar bond and a source of hydrogen bonding, the strongest type of intermolecular force. Water has two of these polar bonds making it an extremely polar molecule. Hydrophilic, “water-loving,” compounds have polar regions with partial positive and negative charges that are attracted to the polar regions of the water molecule. Common chemical structures leading to hydrophilic nature include hydroxyl groups (-OH), amino groups (-NH
2
), and carboxyl and carbonyl groups (-CO and –COO). Hydrophobic compounds have nonpolar chemical structures and thus are “water-hating,” or not attracted to water. These compounds often have C-C bonds and C-H bonds. It is possible for a compound to be large enough to have both a hydrophilic and hydrophobic region enabling it to have unique properties and functions.
Teaching Strategy: “Molecular dating game” Modified from Dr. Andrew Miranker
In order to help students identify sources and strengths of attraction, assign each student a more complex molecule. The twenty amino acids are great molecules to use, as they have sources of hydrogen bonding, hydrophilic and hydrophobic regions, and charges. Ask students to identify their molecule in the following categories: hydrophobic, hydrophilic, or neither; positively charged, negatively charged, or neither; short, medium, or long; and bulky or linear. Ask students to “speed date” with at least three other classmates to find the best match. In each “date”, students should record the other person’s information and conclude what type of intermolecular forces and interactions they had between each other. Good matches could be between two hydrophilic molecules, two hydrophobic molecules, two molecules with long chains for van der Waals forces, or a negatively charged molecule and a positively charged molecule.
Unique Shapes and Functions – An introduction to the chemistry of textiles
The fourth part of this unit will be exploring the field of textiles and uncovering the molecular structures that give rise to each textile’s unique properties and in turn its use in everyday life. Students will be given the shape and component elements of each textile molecular unit and be challenged to find the chemical reasoning behind the textile’s function by creating a model of the molecular unit and exploring it through physical manipulation.
Textiles are an ancient and crucial part of our society dating back to the use of flax in Egyptian linen and silk in Chinese fabrics. Today, the textile field is expanding rapidly beyond just use in clothing and coverings. Through the use of chemistry, textiles are advancing to meet the needs of consumers from fabrics that allow people to wear technology to stain and water resistant clothing to flame-retardant space suits.
A textile is defined as a material that is made of interlacing fibers (thread or filament). A fiber is usually many polymers that are arranged and combined in a variety of bonding and non-bonding interactions. The method of interlacing fibers and the type of fiber used gives rise to the nearly endless variety of textiles available today. The method of combining fibers ranges from knitting to crocheting to blending and everything in between; these methods go beyond the scope of this unit. The type of fiber used can be categorized into two main categories: natural and synthetic. Natural fibers include fibers found in animals, plants, and minerals. Silk, wool, cashmere, cotton, hemp, bamboo, asbestos, and glass fiber are a few popular examples. Synthetic fibers refer to fibers that are man-made, sometimes to imitate the qualities of natural fibers at a lower cost or to create textiles with properties not found in natural fibers. A few common examples are nylon, spandex, polyester, carbon fibers, and aramid.
Teaching Strategy: Exploring Fabrics
Before beginning content on textiles and properties, bring in swatches of different fabrics either in the form of clothing or in fabric swatches; most science kit companies have test fabrics for dyes. It is reasonable to find clothing that is 100% cotton, polyester, nylon, silk, linen, and spandex. Additionally, you can challenge students to bring in a piece of clothing that they particularly like for its texture. Ask students to feel and make observations about each fabric and highlight major differences between the samples. Afterwards, ask students to hypothesize what makes these fabrics so different from one another and generate questions the class would like to explore on a large poster or on the board.
Significant Organic Functional Groups in Textiles
Before diving into the chemical structures that contribute to different textile properties, it is useful to review common functional groups in the fibers that will be referenced. Refer to figure 1 as you read the next few sections.
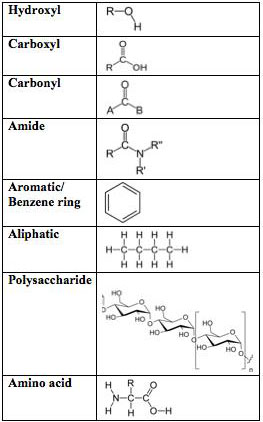
Figure 1
Properties of textiles
The properties of a particular type of textile depend heavily on the structure of its individual fibers, which goes well beyond the simple arrangement and bonding of the atoms that make up the molecules of the fiber. The properties of synthetic fibers also depend on the formation process of spinning, hot drawing, stress relaxation, and heat setting (Hegde, et al. 2004), all which go beyond the scope of this unit.
We will begin by discussing various properties of textiles and the chemical structure that enables them to exist and finish by comparing and contrasting the most prominent textiles in each category, exploring each textile’s basic chemical structure, properties, and the connection between the textile’s form and function. Some textile properties can be easily explained using chemical structure, while others depend on much more complicated macrostructures and on the process of creating the fibers themselves which we will not discuss.
When a textile stretches, its individual polymers can be sliding past one another, requiring the polymers to have loose interactions between chains or parts of the polymer can be unfolding, requiring the polymer to have side chains or branches that fold. Loose interactions between polymer chains indicates that the chemical structure of the fabric’s polymers lack regions of polarity, most likely containing aliphatic sections. A fiber with folding and unfolding branches is more likely to be elastic, returning to its original shape instead of remaining stretched out.
The ability of a textile to be dyed depends on how well the molecules of its fibers can interact with the dye molecules. For example, a textile with hydrophilic properties can be dyed by a hydrophilic dye because they are attracted to one another. A hydrophilic fiber must have some kind of polar region, an area where two atoms of very different electronegativity are bonded and are unevenly sharing electrons to create an uneven distribution of charge. Sometimes, fibers are stretched when dyed to allow dye molecules to get in between fibers. After the fibers are relaxed, dye molecules are trapped between fibers.
Softness requires that fibers have a fine diameter while fluffiness requires molecules that are crimped and can trap air. Softness indicates that the fabric can be easily deformed, which means at a molecular level that polymer chains are easily sliding past one another because of loose interactions between chains. Smoothness comes from a flat, planar structure of molecules and a rough, textured surface would have molecules that are not arranged in orderly sheets but instead may have many side chains and branches that do not evenly stack. Textiles made of long filaments instead of short, staple fibers also contribute to a smooth texture.
The strength of a textile is dependent on the rigidity of its structure and the intermolecular forces between fiber molecules. A fiber with a highly ordered structure is considered to have high crystallinity and the higher the degree of crystallinity, the stronger the material and the less able it is able to elongate before breaking. A structure with double or triple bonds or benzene rings is rigid and able to maintain its form under pressure, while aliphatic compounds with more flexible single bonds are much more likely to bend or break under pressure. Fiber molecules with polar regions, particularly H-O or H-N bonds, are able to hydrogen bond with adjacent molecules, keeping molecules of the fiber close together and contributing to the strength of the textile. Finally, fibers whose molecules are able to stack are able to increase van der Waal forces between chains, also increasing strength.
To be absorbent or wicking, a fiber must have hydrophilic molecules, a molecule with regions of polar bonds, especially N-H and O-H bonds. High absorbent capacity requires that the textile have sufficient space between fibers to hold molecules. To be water resistant, a fiber’s molecules must be hydrophobic, having nonpolar bonds. Some fabrics are absorbent, like cotton, but water molecules will accumulate in the fabric, preventing evaporation into the air; this is especially undesirable when the trapped molecules are sweat. In wicking fabrics, like dri-fit, polyamide groups draw molecules from the skin through the fabric to the outer layer where they can be evaporated instead of making the fabric wet and smelly. Polyamides are polymers with polar groups on both ends that are very attracted to water molecules and a highly organized crystalline structure in the middle. The watery, sweat molecules are pulled into the fabric by their attraction to the outer polar groups and then pulled through the fabric to polar group on the other end where they are evaporated and are unable to penetrate and get stuck in the crystalline structure in between. (Peso 2007).
Breathability of fabrics refers to how well fabric is able to allow moisture vapor to pass through instead of getting trapped, known as moisture vapor transfer (MVT). Fabrics with hydrophilic molecules are able to move water molecules through the fabric by Coulombic attraction between the polarity of the water molecule and the polarity of the fabric molecule.
Teaching Strategy: Tests for properties
While exploring the properties of different textiles, student can test the stretch, dyeability, and absorbency of different fabrics. For stretch, ask student to equally stretch each material and visually observe which material stretches the farthest. Also, observe which fabrics are able to return to original form, noting this property as elasticity. Contrast stretch with elongation to break by having students measure how far they can stretch a material before it breaks. To test for dyeability, use the test fabric from a science kit company using different kinds of dye. And for absorbency, lay different swatches of fabric on the same amount of water and qualitatively observe how much water is left behind when the fabric is removed after five minutes.
Teaching Strategy: “Which molecule is best”
When detailing the connection between structure and properties, present several unlabeled molecules of fabrics and ask students to choose the molecule that would best display each property and explain why. See figure 2 for examples to use.
Natural Fiber Examples
Cotton is perhaps one of the most recognizable fabrics in clothing. It is comfortable, easily dyed, and a relatively tough textile, able to withstand frequent washing and wear. It is produced from the seed of a Gossypium plant and is primarily composed of cellulose, (C
6
H
10
O
5
)x, a polysaccharide. It contains two six-membered rings joined together by an oxygen atom (see figure 2). Cellulose has three hydroxyl groups (-OH) leading to extensive hydrogen bonding between cellulose chains and giving it hydrophilic properties and high reactivity to a variety of compounds. This reactivity allows cellulose to easily interact with many dyes as well as blend easily with others fibers to create new fabrics with enhanced features. The hydrophilic nature of cellulose also gives it high absorbency in water leading to softness and comfortableness. However, during water absorption, polymer chains in cellulose are strained and shift to reduce this stress leading to new hydrogen bonds manifesting macroscopically as creases in the fabric. Despite the branching present in cellulose, the molecule is fairly flat allowing the chains of cellulose to easily stack and form a highly ordered crystal structure. This crystal-like structure gives cellulose low elasticity, leading to stretched-out fabrics after extensive wear. (Hegde, et al. 2004) (Mather and Wardman 2011).
In contrast to the plant fiber of cotton, wool is made from much more complex fibers of sheep hair. Wool is used for its warmth, softness, and water resistance. The primary fiber of wool is a protein called keratin, but wool also contains wool grease (esters), suint, and cortical and cuticular cells. Keratin is the major protein of our hair and nails and different variations of keratin and other components give rise to different products like wool, cashmere, and mohair. Keratin is made of a variety of amino acid residues but the most significant amino acid influencing its properties is the cysteine residue containing a sulfide bond leading to disulfide bonds linking adjacent keratin chains. These disulfide bonds are significant in that they are actually covalent bonds and not just intermolecular attractions between keratin chains creating a much stronger link between keratin chains. This strong bond leads to high elastic recovery, which is the ability to retain shape after being stretched out. (Mather & Wardman, 2011). Crosslinking cysteines in human hair are the source of curly or wavy hair. When hair has a perm or is straightened, these cysteine-cysteine bonds are broken and then reformed with new combinations when the hair has the new desired shape or texture. The natural curl of wool due to cysteine bonds enable the fibers to trap air, giving wool its ability to retain heat and feel comfortable.
Another type of textile made from protein fibers is silk, produced from the bombyx mori larvae feeding on mulberry leaves or from spiders. Silk is a luxurious fabric praised for its smooth feel and strength. The primary protein in silk is fibroin, composed of several amino acids, mostly glycine, alanine, and serine. In contrast to wool, fibroin does not contain the major amino acid, cysteine, as bombyx mori or spiders have no need for the advantages that curly fibers bring. These three primary amino acids also have the smallest side chains, allowing the fibroin chains of silk to stack neatly on one another. This planar structure leads to the smooth surface of silk. Silk is very strong compared to wool, which can be attributed to the strong intermolecular forces between the stacked, planar sheets. However, wool is much more stretchy than silk whose amino acid residues are already extended while wool has longer side chain which can fold and unfold. (Mather & Wardman, 2011) (Humphries 2009)
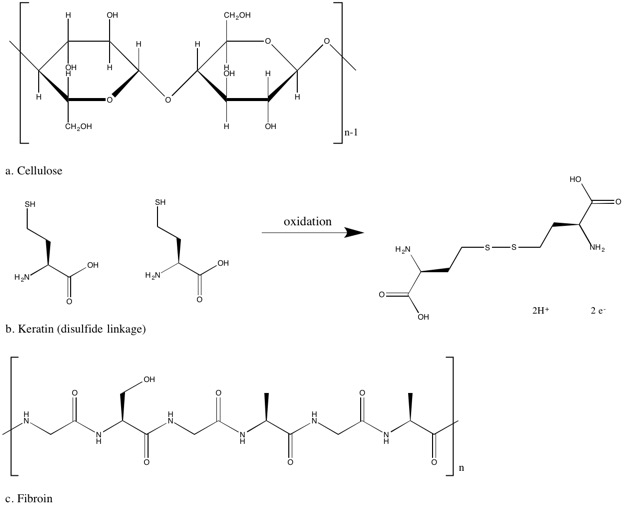
Figure 2
Synthetic Fiber Examples
While natural fibers offer seemingly endless options for textiles, the demand for synthetic fibers has surpassed natural fibers since the 1990s. Synthetic fibers are produced from nonrenewable, polymer granules, often petroleum, that have been melted or made into solution and made into continuous filaments to be spun into fibers. The production of synthetic fibers began in the 1930’s in attempt to mimic the properties of natural fibers at lower cost and to produce new fabrics with different combinations of properties than what could be found in natural fibers. Many synthetic fibers have lower glass transition temperatures (where substance becomes rubbery), the point at which bonding between chains disappears. This allows dye molecules to get between chains and when the fiber is re-cooled, the dye molecules are effectively stuck between chains. See figure 3 for images of the prominent molecules of synthetic fibers.
Nylon, a polyamide, was the first synthetic fabric produced and was made with the goal of imitating silk. It quickly found great use in pantyhose, brushes and bristles, fishing lines, and parachutes. Nylon has several conformers, each containing C-C bonds, carbonyl groups, and several N-H bonds. Nylon is extremely strong having a tensile strength per unit weight stronger than steel wire (Lancashire, 2015) thus leading to its widespread use in the automobile industry in materials like tire cords. Because nylon has chains that are largely aliphatic, these chains are able to stack close together through van der Waals forces. This close stacking enables hydrogen bonding between C=O and N-H in neighboring chains further keeping the chains of nylon together and leading to its stiffness, resistant to tear, and high strength. (Dasgupta 1996) (Hegde, et al. 2004)
Polyester has become an extremely widely used fiber in everything from clothing to ropes and often in a blend with other fibers. The most prominent polyester is poly(ethylene terephthalate) (PET). In the repeating linear unit of polyester there is an aromatic ring attached on either side to carbonyl group and extended on one side to two CH
2
groups. The structure of PET has both crystalline and noncrystalline regions. PET is resistant to acids and many solvents but susceptible to alkalis. Alkalis are able to attack PET through its noncrystalline regions creating a loss of properties and introducing a porous surface, which is sometimes used to enhance the fabric (Hsieh 1996). The aromatic ring adds rigidity to PET’s structure and prevents polyester from deforming easily which is seen at the macroscopic level as wrinkle-resistant. This rigidity and crystalline regions also prevent PET from absorbing dyes. Consequently, a large amount of research has been carried out to find ways to dye polyester by polymerizing it with a third monomer that has reactive dye sites (Pal 1993). Polyester also has a fairly high tenacity, which can be attributed to the hydrogen bonding and van der Waals force between chains (Hegde, et al. 2004).
Next, a rising category of synthetic fibers is performance fibers including spandex (Lyra or elastane) used for its incredible ability to stretch and prevalence in countless athletic wear. Spandex is a polyurethane molecule containing rigid and flexible segments that allow it to be both tight and stretchy. The flexible section is one oxygen connected linearly to a CH
2
group attached to another CH
2
group. All bonds are single bonds granting this section ease of rotation around these bonds. The rigid section contains numerous double bonds in several aromatic rings and carbonyl groups as well as N-H bonds. (Brown 2000)
The last textile we’ll examine is Kevlar, a high strength textile used in a variety of applications like cables, brake linings, bicycle tires, and body armor. In figure 3, you can see that Kevlar has repeating benzene rings connected by amides. These repeating benzene rings give Kevlar a very rigid structure, similar to the rigid parts of spandex. These rigid molecules combine into planar, sheet-like structure that tend to stack one another using van der Waals intermolecular forces (Petty n.d.) This rigidity throughout the Kevlar structure as well as the hydrogen bonding between oxygen and hydrogen atoms of adjacent chains gives it incredible strength five times stronger than steel (Dupont 2015). Kevlar is cut, chemical, flame, and slash resistant
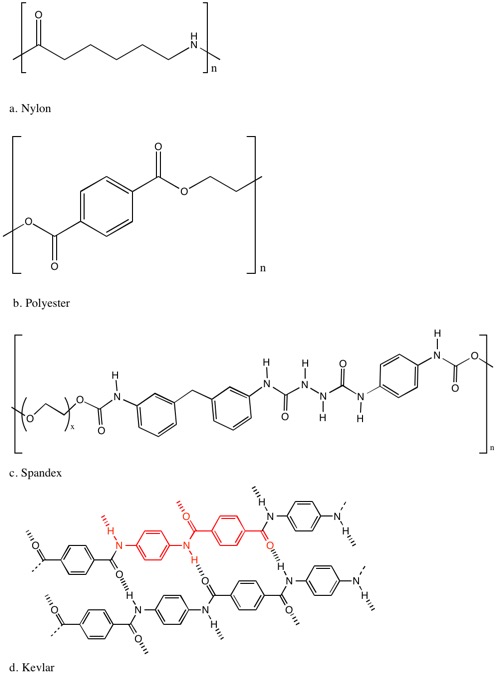
Figure 3
Project assessment
The final part of this unit will be exploring ways to visualize and model molecular shapes. Students will use simple tools like Lewis dot diagrams and VSEPR as well as advanced computer software like Pymol. The unit will culminate in a molecular modeling project with students choosing a textile molecule to model and preparing a poster to explain what forces influence its shape and how its structure determines its function and properties. A student-ready and formatted handout follows below.
Molecular Modeling Project
In this project, students will be choosing a common molecule and exploring why its size and shape are important to everyday life. You will explain the everyday implications of this molecule’s structure to your classmates by designing a 3D model of this molecule and a poster detailing its properties and unique structural features that cause these properties.
You will be assessed individually on your model, your poster or PowerPoint presentation, your ability to explain the connection between your molecule structure and its properties, and your discussion and reflection of your classmates’ projects.
Here’s a great example:
Carbon monoxide (CO) is a small molecule that exists as a gas at normal temperatures. Carbon monoxide is one of the most common types of air poisonings if inhaled in concentrated amounts. It is often produced by incomplete burning of fossil fuels and can come from tobacco smoke or defective central air systems, stoves, or space heaters.
|
Molecular mass
|
28.010 amu
|
|
Density
|
1.145 kg/m
3
|
|
Melting point
|
-205.02
o
C
|
|
Boiling point
|
-191.5
o
C
|
|
Solubility in water
|
27.6 mg/L
|
The reason carbon monoxide can be toxic is because its molecular structure is very similar to oxygen and if inhaled, it can bind with hemoglobin instead of oxygen leading to hypoxia (lack of oxygen). Both molecules consist of two atoms of very similar size and electronegativity and both molecules have a linear shape and short bond length because of their double/triple bond.
The bond between carbon and oxygen in CO is a relatively nonpolar bond because carbon and oxygen have very similar electronegativity values. Because water is polar, CO does not have a high solubility in water due to the lack of attraction between water molecules and CO molecules. The lack of polarity in CO also means that its melting and boiling point are pretty low because CO molecules are not attracted to one another, also explaining why CO is found as a gas at normal temperatures.
-
Find out its properties!
Here’s brainstorming questions to get you started in your exploring:
-
What is this textile used for?
-
What unique properties does this textile have?
-
Why is this textile important?
-
What should others know about this textile?
-
How is it made?
-
Where is it found?
-
Pick a textile you’re interested in!
Here’s some examples listed from easy to hard. Be sure to get your choice approved
-
Cotton
-
Linen
-
Hemp
-
Silk
-
Spandex
-
Nylon
-
Polyester
-
Model it!
-
Show accurate angles
-
Show Polar and/or nonpolar bonds
-
Use Labels for ALL atoms
-
You can use any of the following materials
-
3D drawing
-
gumdrops and toothpicks
-
clay - extra credit
-
styrofoam balls and wood sticks - extra credit
-
anything you can think of! Get it approved by Ms. L
-
Communicate your information to your classmates!
-
Use a poster or PowerPoint presentation
-
Be sure to include at least the following properties
-
Melting point
-
Boiling point
-
Solubility in water
-
Density
-
Include an explanation of how the structure of the molecule relates to its properties
How to Explain Your Molecule’s Properties
Compared to water, the melting and boiling point of my substance is __________________ (high/med/or low). My substance is a ________________ (solid/liquid/ or gas) at room temperature. In order for a substance to be a solid, its individual molecules (which I made a model of) must stick together and be very attracted to each other. In order for a substance to be a gas at room temperature, its individual molecules must be very spread apart and not attracted to one another. Attraction in molecules only happens between (+) and (-) so if my substance is a liquid or solid at room temperature, they must be some positive and negative parts in the individual molecules of my substance. Positive and negatives areas can develop from unequal sharing of electrons between atoms. Some atoms want electrons more than others; this desire for electrons is called electronegativity and is measured on a scale from 0.0 to 4.0. If atoms are bonded but one atom has a much higher electronegativity than the other atom connected to it, the electrons will stay closer to the more electronegative atom, making the part of the molecule negative and the other atom more positive. When these positive and negative parts develop, the individual molecules are more attracted to each other, remaining as solids and liquids for longer periods of time. This makes the substances melting and boiling points higher. The more polar regions a molecule has, the higher the melting point and boiling point will be. Polarity in a molecule also leads to greater solubility in water because water molecules are polar and will be attracted to the molecule and surround it, dissolving it.