This initial subunit addresses the background information necessary to understand the anatomy and physiology of bacteria and viruses. At the end of this subunit, students should be able to identify the major structures of bacteria and viruses and the differences and similarities between them.
Bacteria Anatomy, Morphology, and Arrangement
In this portion of the subunit, students will learn about the basic anatomy and structures that are common with bacterial pathogens. In addition, students will learn about differences in the peptidoglycan cell wall and capsule of bacteria species and how this structure is important for identifying the bacteria. Students will also learn basic morphology and arrangement of bacterial species, a key feature to identifying the infectious agent. It is recommended that students use the biology texts issued by their districts or sample microbiology text books to assist them with this portion of the unit.
Bacteria are microscopic, prokaryotic organisms known to cause several debilitating, contagious diseases. Bacteria are ubiquitous and, while often depicted in a negative light by media, are not all harmful. Most living organisms live in synergy with bacterial cells and receive many benefits from their mutual interaction. Humans are covered in native flora which provides us with several important health benefits.
24
However, not all bacteria are beneficial. Several bacterial species are known to cause debilitating or deadly diseases.
Bacteria possess many of the same cellular traits that students are familiar with from introductory biology units, however, it is worth reviewing the function of basic organelles. Students should also be reminded of the meaning of prokaryotic and the key differences between this formation of cells and the eukaryotic cells. There are also parts of bacteria cells that students may not have been exposed to in prior units (see Figure 1).
In addition to the cell membrane, bacteria possess two additional external structures called the cell wall and the capsule. Students may be familiar with the concept of a cell wall if they have already studied plant cells. It is important to note that the bacterial cell wall is made of a substance called peptidoglycan, not the cellulose found in plants, and is found in one of two types. Bacteria with a very thick cell wall are considered to be Gram Positive, while thin cell walls indicate Gram negative species.
24
The cell wall provides several advantages to the bacteria, including the regulation of turgor pressure from interactions with the environment.
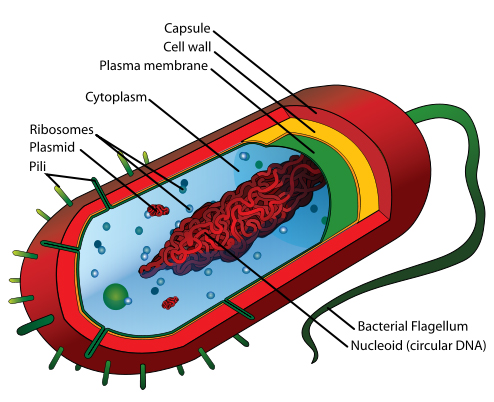
Figure 1. A sample prokaryotic cell with major parts labeled. Retrieved: http://en.wikipedia.org/wiki/Bacteria#mediaviewer/File:Average_prokaryote_cell-_en.svg
The cell capsule is found mostly in Gram negative species, although there are a few notable Gram positive species that do have one present. The cell capsule is a virulence factor in most pathogenic bacteria species.
24
This structure prevents phagocytosis and desiccation of the bacterial cell and allows it to adhere to surfaces aiding in the formation of biofilms or fomites.
24
Additional external structures of bacteria include the pili—short hair like structures found projecting outward that assist in movement through a rowing motion—and the long whip-like flagella. Students should become familiar with several flagella arrangements, including peritrichous, lophotrichous, and amphitrichous morphologies.
24
Some bacterial species can form structures called endospores. Endospores are resistant encapsulations that allow for bacteria cells to survive hostile environments. Endospores are not reproductive, but may allow a bacteria to survive in an otherwise dangerous environment until conditions become suitable for it to reactivate into a vegetative or reproductive state.
24
There are even cases of endospores being revived after millions of years of hibernation.
2
Students should also be exposed to the three common morphologies of bacterial cells—cocci, bacilli, and spirillum. In addition to being able to recognize common morphologies, students should also be familiarized with the arrangements that these forms take. Specific morphologies have particular arrangements that are common. Bacterial species will favor one arrangement over others and these two pieces of information together can be important in the diagnostic identification of bacteria. Students should be exposed to staining techniques that allow them to identify the morphology and arrangements of certain bacteria. This will allow them to dissect some bacterial names and identify bacteria from slides or to construct what certain bacterial colonies should look like given the name (see Figure 2).
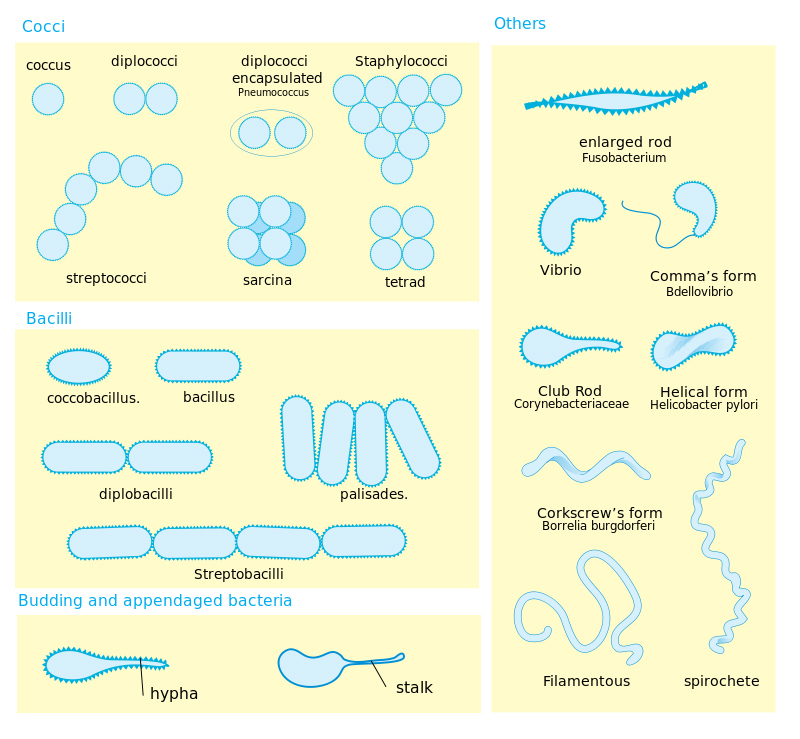
Figure 2. A diagram of bacterial morphology and arrangements. Retrieved: http://en.wikipedia.org/wiki/Bacteria#mediaviewer/File:Bacterial_morphology_diagram.svg
It is important to note, however, that there are several other bacterial morphologies that students might encounter outside of the big three. Coccobacillus bacteria, for example, are more oblong than a normal coccus, but do not have the length associated with the rod shaped bacillus bacteria. Examples of coccobacillus species include the re-emerging bacterium
Bordetella pertussis
, the causative agent of whooping cough.
In addition, I have loosely grouped spirillum into one class when, in reality it should be divided into three subgroups: vibrio, spirillum, and spirochete. Vibrio morphologies, as seen in the bacterium
Vibrio cholerae
are comma or curved rods. These bacteria do not have the exaggerated spiral shape that is often associated with the spirillum grouping. Spirillums are often bacteria that are characterized by thick, rigid spirals. Common examples of this bacterial morphology are
Spirillum volutans
and
S. minus.
The last classification within this grouping is the spirochetes. Spirochetes are often thinner and more flexible than spirillum. They also exhibit a higher volume of curl than is seen in the more rigid, thicker bacteria. Examples of spirochetes include the causative agent of Lyme's disease,
Borrelia burgdorferii,
and
Leptospira.
Bacteria Staining Protocols
Two staining protocols commonly used in microbiology labs to identify bacteria morphology, arrangement and, structure, are simple staining and negative staining. Simple staining is, as the name implies, relatively easy to perform. This type of staining is ideal for a laboratory class and can be modified to suit every facilities particular constraint and needs. Simple stains are performed using one basic (positively charged or cationic) stain such as methylene blue or safrinin.
24
These stains color the cell because they have a slight positive charge to their chromophore—the name for the grouping of the compound that contains the color. Bacterial cells are slightly negatively charged. When introduced to a basic stain like safrinin or methylene blue, the stain readily binds with the cell proteins, essentially staining it the color of the chromophore.
24
As a result, cells stained with basic stains are the color of the dye itself.
It is important to note that heat fixing is crucial for developing a good simple stain. Heat fixing is used to stick bacterial cells to the slide. If this technique was not used, the stained cells would be washed off the slide when the excess dye is removed. Heat fixing requires passing a bacterial smear over a flame. The flame will cause some of the proteins to stick to the slide as they are heated with only a small amount of distortion to the organism.
24
Too little heat, and the cells will wash off the slide. Too much heat will cause a great amount of distortion, drastically changing their perceived morphology and arrangement or destroying the cell entirely.
24
After heat-fixing, students can apply the simple stain of their choice to the slide. The stain will sit according to the protocol, usually around one minute to ensure strong saturation, and then is rinsed off using sterile or distilled water. After, the slide should be blotted with bibulous paper to remove excess moisture. At this point, the slide can be observed.
Negative staining, unlike simple staining, uses dyes that have a negative charge. Many of the stains used in negative staining are also called acidic staining. They have a negative charge to their chromophore.
24
The stain and the cell repel each other. As a result, students are left with a slide that looks like a negative of a film strip—the background is stained black while the cell itself is not. This staining technique is also referred to as indirect staining, because of the way it stains the slide and not the cells. The most common negative stain is nigrosin. Negative staining is an excellent tool for studying morphology and arrangement because no heat fixing is required to set the cells on the slide and so the highest degree of preservation is observed.
24
To create negative stained slides, students will need to add a drop of nigrosin to the edge of one slide. Into the drop, mix a loop of suspended culture and agitate to break up large clumps. Using the end of another slide, students will draw the nigrosin-culture mixture across the side. Slides are usually observed after the film air-dries. It is very important to note that negative stained slides are not rinsed with water after the smearing process. Rinsing with water would remove the dye and the cells, leaving clean slides.
These are just two of the many staining techniques used in laboratory to observe morphology and arrangement of bacterial cells. We will learn another staining technique called Gram Staining in subunit four.
Virus Structure
Viruses, unlike bacteria, are not composed of cells. Instead, they are pieces of single or double stranded DNA or RNA surrounded by repeating protein subunits called capsomeres to form a polyhedral or a helical capsid structure.
24
While many human pathogens are polyhedral virus crystals, it is important to expose students to the multitude of viral shapes including the 'spider-like' T4 bacteriophage shape with tail fibers and tail sheath and the
filovirus
family with their long, Sheppard's crook formations.
Students should also be exposed to the lytic and the lysogenic replication cycles of viruses and how the basic components of virus structure allow it to infiltrate host cells and hijack their cellular machinery (see Figure 3).
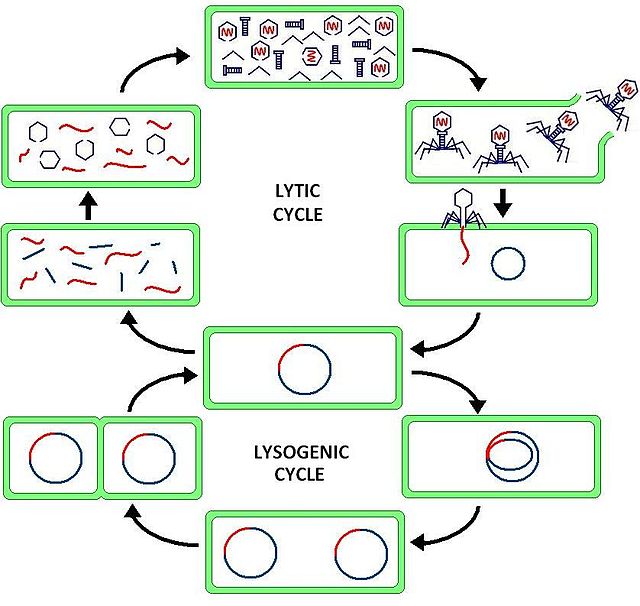
Figure 3. The viral lytic and lysogenic cycles. Retrieved: http://en.wikipedia.org/wiki/Lytic_cycle#mediaviewer/File:Phage2.JPG
Viruses are relatively immobile—they do not actively seek out hosts. Instead, they passively move through the human body until they encounter a host cell with receptor membrane proteins that trigger it to attach. If the virus is a bacteriophage, it will inject the genetic information directly into the host cell. Some enveloped viruses have similar receptor molecules and will "merge" with the host membrane as if it were a liposome, entering the bacteria at that point.
24
Once the viral information enters into the host cell, it will hijack the ribosomes and synthesize viral proteins in a replication process. The viral particles self-assemble inside of the host cell and will lyse or burst out upon completion. If the virus is an envelope virus, it will cloak itself in the cell membrane of the host as it rips its way out.
The lysogenic cycle follows the processes of attachment to the host cell and injection, but once inside the cell, the viral genetic material inserts into the host cell's DNA. This stage is referred to as the formation of a prophage. Prophage refers to the combination of the viral and host genetic information.
24
At this point, the virus enters a latent stage where the host cell replicates, incidentally replicating the viral material as well. This latent, shadow stage can last several weeks to years as is seen in the case of the HIV retrovirus. As the cells proliferate, so does the viral material until there is a chemical signal that causes it to undergo spontaneous induction. At this point, the viral DNA exits the host DNA and enters into the lytic cycle.
There are several major differences between the lytic and the lysogenic cycles. They lytic cycle, while rapid, affects one cell at a time. While the number of cells infected increases with the viral load, there is a noticeably logistic growth to this type of infection within the host body. They lysogenic cycle will infect one cell and then 'hibernate', taking a long time for this cycle to undergo completion. However, the virus passively replicates as host cell turnover can, in effect replace a large amount of the cell tissue with the newly infected cells as they proliferate. Then, once spontaneous induction occurs, multiple cells burst releasing a dauntingly large viral load into the host.
Recommended teaching strategies for this subunit allow students to explore the structures of viral and bacterial pathogens and the important difference between them. One recommended teaching strategy is to allow students to study preserved slides of various bacteria and identify their morphology and arrangement. Students will have the added advantage of learning to focus microscopes under oil emersion (100x oil emersion objective lens) and manipulating sides on a light microscope. It is also recommended that students be allowed to build model viruses and label the structures.