Terry M. Bella
The phenomenon of ocean acidification refers to the rapid increase in acidity of the world’s oceans. When ocean pH is discussed it is in reference to a global average ocean pH. It should be noted that the pH of seawater is not consistent spatially across the globe. There are areas with significantly higher and areas with significantly lower pH values. Since the year 1760, the start of the industrial revolution, the average ocean pH has dropped from 8.16 to 8.07. This can be attributed to the carbon dioxide that is continually emitted through the combustion of fossil fuels. Carbon dioxide readily dissolves into water as it attempts to equilibrate concentrations between the ocean and the atmosphere. Based on atmospheric CO
2
measurement and Henry’s constant for CO
2
, it is estimated that the ocean absorbs approximately 26% of anthropogenic carbon dioxide which equates to 2.5 billion tons of the gas annually. Estimates are drawn from data collected between 2001 and 2011 (4). Henry’s law describes the amount of a given gas that will dissolved in solution determined the by the partial pressure of the gas in the system.
Dissolved carbon dioxide (CO
2
) chemically reacts with water (H
2
O) to form the weak acid carbonic acid (H
2
CO
3
). Being that H
2
CO
3
is an acid it dissociates in water forming hydronium ions (H
+
) and carbonate ions (CO
3
2-
), pK
a
’s for this divalent acid are 6.3 for bicarbonate (HCO
3
-
) and 10.3 for carbonate (CO
3
2-
). This initially seems like a benefit for shell building organisms because they utilize CO
3
2-
, combining it with calcium ions (Ca
2+
), to form calcium carbonate (CaCO
3
) the main constituent of the shell. Unfortunately, excess of H
+
in the water compete with Ca
+2
forming bicarbonate (HCO
3
-
), effectively locking up the CO
3
2-
. Furthermore, carbonate availability decreases as pH decreases. Below, the ionization fraction plot of carbon dioxide (see Figure 1). Note how the equilibrium between ionic states is pH dependent and as we drive the pH of the ocean down, available carbonate decreases. The pK
a
is an equilibrium constant indicating the pH were 50% of a compound exists in the H
+
associated form (e.g. HCO
3
-
) and 50% exists in the H
+
dissociated form (e.g. CO
3
2-
). Not all organisms rely on free carbonate, but those that do will have increasingly difficult time acquiring free carbonate in the seawater as the ocean becomes more acidic. Below are the chemical equations for the formation of CO
3
-2
(see Figure 2). These free H
+
are the “acid” in acidity, the more H
+
there are in solution the more acidic the water is. The more CO
2
there is in the atmosphere the more CO
2
there will be dissolved in the ocean, resulting in free H
+
.
Figure 1: Graphical representation of the pH dependent ion concentrations of carbon dioxide in water. Note that with a pH below 8 concentrations of carbonate are negligible. (5)
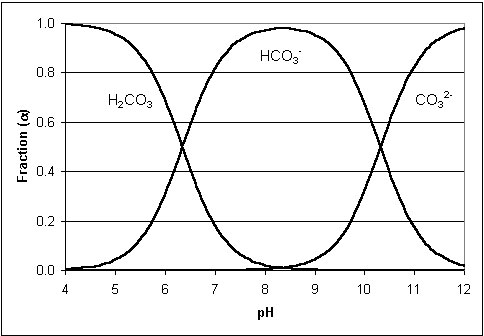
Ionization fraction plot of carbon dioxide.
Figure 2: Series of chemical reactions involving carbon dioxide and water.
C0
2
(g) + H
2
O ⇄ H
2
C0
3
(aq)
H
2
C0
3
(aq) ⇄ H
+
(aq) + HCO3
–
(aq)
HCO3
–
(aq) ⇄ H
+
(aq) + CO
3
2-
(aq)
Chemical reactions of carbon dioxide in water.
The pH scale runs from 0, strong acids, to 14, strong bases (see Figure 3). A pH reading is an indication of the concentration of hydronium ions (H
+
) in solution. The p denotes negative logarithm base 10. The higher the pH value the less free hydroniums and conversely the lower the pH the more free hydroniums there are. The scale is also logarithmic and each movement from one number to another in sequence represents a 10 fold difference in hydronium concentration. For example, the concentration of hydronium ions in a solution of pH 9 is ten times higher than that of a solution with pH 10. A pH value of 7 is neutral. Readings above 7 are in the basic or alkaline range of pH, see Figure 3 below. The ocean, because of all the dissolved minerals that buffer the water, has historically had a basic pH. Throughout this unit I may interchangeably refer to the pH of the ocean becoming “more acidic,” “less basic,” “less alkaline,” or that it is “losing alkalinity.” There is some confusion around the term “ocean acidification” when the ocean is not in fact acid to begin with and has not become acidic, it has become less basic. Ocean acidification is so named to describe the addition of carbonic acid (H
2
CO
3
) via the dissolution of CO
2
gas.
Figure 3
The pH scale.
Connecting the Carbon Cycle
The advent of the industrial revolution has included the widespread and massive use of fossil fuels. Fossil fuels are coal, oil (including derived products), and natural gas. Coal was the primary source of energy originally, followed by advent and inclusion of oil, and then natural gas. These energy dense, due to their hydrogen-carbon bonds, materials were vital to the modernization of the world. The combustion of these carbon based fuels, as well as land use changes, releases CO
2
in the atmosphere. In 2016 36,183 metric tons of CO
2
were emitted (6). This is equivalent to 79,769,765 pounds of CO
2
. Considering that the ocean absorbs 26% of the anthropogenic CO
2
, over 10,121 metric tons CO
2
were dissolved into seawater in excess of natural sources.
Carbon cycles through this planet, from reservoir to reservoir, through a set of biogeochemical processes. Reservoirs can be differentiated as long-term or short-term based on the relative amount of time that any given carbon atom will spend within them. Long-term reservoirs refer to processes or sinks that sequester carbon for tens of thousands to millions of years. Examples of long-term reservoirs include rock, such as limestone, fossil fuels, and the deep ocean. Conversely short-term carbon reservoirs allow carbon to move in and out of them in time spans as short as minutes to thousands of years. Examples of short term reservoirs include all living things, the atmosphere, and the shallow ocean. The vast majority of carbon on earth is locked up in long-term storage, primarily limestone formations. There are some 100,000,000 GT (gigatons) of carbon sequestered in limestone (7). Consider that the estimated total carbon on this planet is roughly 100,050,000 GT. The carbon in the fossil fuel derived carbon dioxide that is decreasing the pH of the ocean was in long-term storage, until it was removed by humans. It can be argued that carbon is simply being cycled through it’s natural set of reservoirs and will simply keep cycling. However, humans have accelerated the cycling, and changed balances by moving gigatons of carbon out of long term storage in fossil fuels, releasing them into short-term reservoirs, the atmosphere and shallow ocean. Through concrete making humans have also moved carbon from long-term to short-term reservoirs. Our activity has increased the amount of carbon in the atmosphere and ocean significantly in a very short amount of time. The excess carbon in the atmosphere is trapping heat, resulting in climate change and the excess carbon in the ocean is driving down the ocean’s pH.
The movement of carbon from the short-term reservoirs and back into the long-term storage is an important solution in correcting the aforementioned problem we have caused. This process naturally begins simply with our shell forming organisms, the bulk of which are living in the oceans. These organisms take carbon, as carbonate (CO
3
2-
) out of the seawater and incorporate it into their structures. Shell, and carbon skeleton forming, organisms include but are not limited to phyto- and zooplankton, crustaceans, bivalves, corals, echinoderms, and mollusks. When these organisms die, their shells deposit on the ocean floor. These calcium carbonate structures accumulate over time and with pressure from the overlying water and layers of sediment are formed into limestone. This process is important because it is cycling carbon out of short-term storage where it affects the chemistry of the ocean or the global temperature and puts it into long-term storage. Thus, beyond a decrease in pH, ocean acidification may inhibit a central, natural mechanism for moving carbon into long-term storage.