Carbon is a nonmetallic element that is incredibly important to many processes on earth, as it serves as the backbone for many living organisms and has the ability to store energy in its bonds. Carbon has 6 protons, and has isotopes with 6, 7, and 8 neutrons. A neutral carbon atom has 6 electrons, 2 in the inner shell and 4 in the outermost shell. It can lose 4 electrons to have a charge of +4 (oxidized cation) or gain 4 electrons to have a charge of -4 (reduced anion). Because of its ability to both lose and gain electrons, carbon can form bonds with many other elements, making it extremely important to many biological and geological processes.
There are four types of carbon molecules that are found on Earth. Organic carbon, like hydrocarbons and carbohydrates, is formed by photosynthesis and makes up living and dead material. It is in a reduced state, so therefore it stores energy. Bicarbonates and carbonates are forms of inorganic carbon that are present as dissolved ions, particularly in the oceans. Carbonate can also be found as a solid inorganic compound in soils, ocean sediments, and glacial flour, finely ground sediment. Finally, carbon dioxide (CO2) is a gaseous form of inorganic carbon that can be found in the atmosphere, water, and soil. Oxidized carbon, such as carbon dioxide and calcium carbonate are the most stable forms of carbon.
Carbon cycle - pre anthropogenic
Carbon naturally cycles through organic and inorganic forms. There are four main reservoirs where carbon is stored. Carbon is measured in gigatons, which is equivalent to one billion metric tons. The atmosphere holds 700 Gigatons (Gt) of carbon in the gaseous form of carbon dioxide. Carbon dioxide makes up a very small percentage of the gases in the atmosphere, only 0.039%. Ocean waters hold 38,000 Gton of oxidized inorganic carbon, 600 Gt of dissolved organic carbon, and 1 Gton of carbon in living organisms. The land holds 500 Gt of carbon in living organisms as well as 1500 Gt of dead carbon in the soil from the decomposition of plants and animals. Most of this carbon (about 1000 Gt) is stored in the permafrost. The concentration of organic carbon in soil in a specific location varies widely depending on the climate and land use. Finally, sedimentary rock holds 1.2 million Gt of carbon in the form of rock carbonates (mainly CaCO3) and 5000 Gton in the form of fossil fuels.2
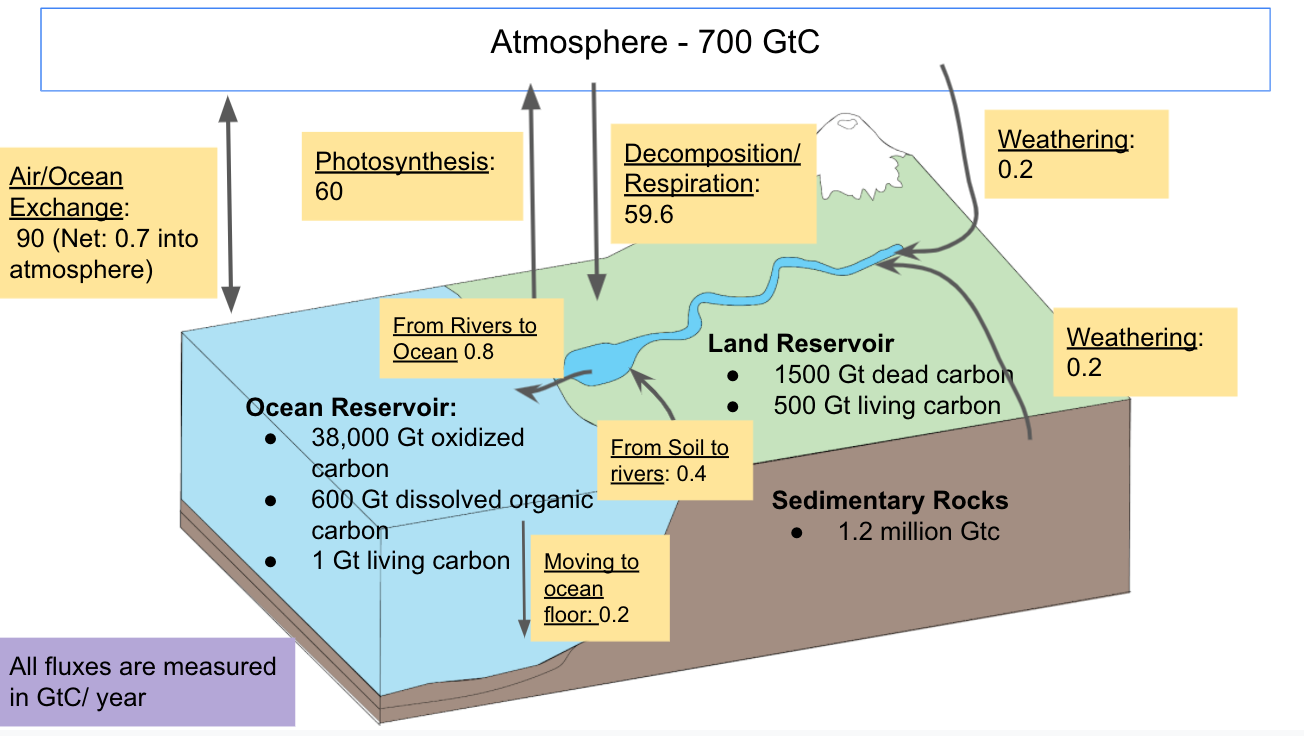
Figure 1: This figure shows the carbon held in each of four reservoirs: atmospheric, ocean, land, and sedimentary rock. It shows the movement of carbon between reservoirs in the preanthropogenic era.
Carbon moves through these reservoirs through three major chemical reactions. The first is that of photosynthesis/ cellular respiration: carbon dioxide reacts with water and energy to produce CH2O and oxygen. In this reversible reaction, oxidized carbon (CO2) is reduced using energy from the sun to create organic carbon. This organic carbon is produced by plants and some autotrophic bacteria on land and in the sea. In the reverse direction, organic carbon is oxidized using O2 in order to produce energy. The by-products of this reaction are carbon dioxide and water vapor. Because of the nature of this reversible reaction, the amount of carbon dioxide in the atmosphere increases and decreases on a yearly cycle. During the growing season, the plants in the terrestrial biosphere use a greater amount of carbon dioxide, causing the atmospheric level to decrease. In the winter, however, the respiration reaction dominates, causing the level of carbon dioxide to increase.
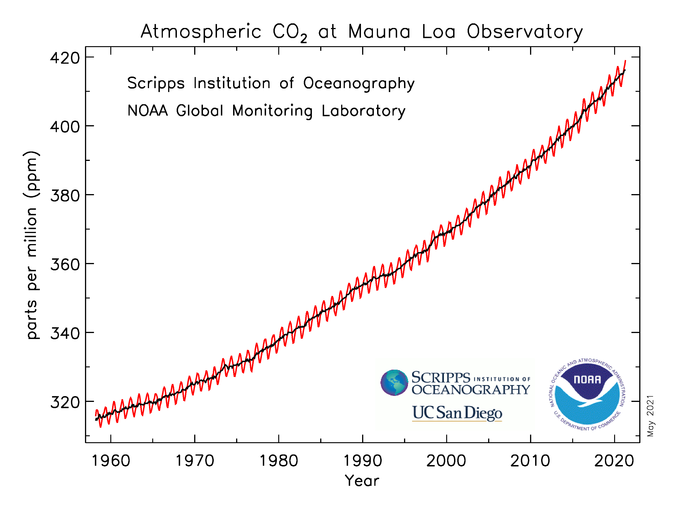
Figure 2: This graph shows carbon dioxide levels measured at the Moauna Loa Observatory in Hawaii. Each year the CO2 decreases in the summer and increases in the winter, causing a cyclical pattern (Source: https://gml.noaa.gov/ccgg/trends/)
On average, 60 Gt is taken out of the atmosphere every year through terrestrial net primary production (reaction 1) and 59.6 Gt is put back in by decomposing organisms on land (reaction 1 reverse).3
The second reaction that moves carbon through the three main reservoirs is a chemical weathering reaction. The chemical reaction for carbonate weathering is calcium carbonate reacts with carbon dioxide and water to form calcium ion and hydrogen carbonate. In carbonate weathering, carbon dioxide from the atmosphere reacts with water to form carbonic acid (H2CO3). The carbonic acid dissociates, allowing hydrogen ions to move freely in the water. These hydrogen ions then react with minerals, such as calcium carbonate (CaCO3) forming clays and soluble ions. The dissolved sedimentary rocks are converted into solids by shell building organisms, like coral and plankton, which form shells from the material. Over time, when the organisms die, they sink to the ocean floor and form sedimentary rock. This is a reversible reaction, so when organisms form calcium carbonate shells, they release carbon dioxide.
The forward reaction is favored in warm, wet conditions on the Earth’s surface. It also occurs faster when the level of CO2 is higher. The reverse reaction occurs in the high temperature and high pressure environment of the Earth’s interior and is called metamorphic decarbonation. The CO2 produced by the metamorphic decarbonation is eventually released back into the atmosphere through volcanic degassing. Unlike the photosynthesis/cellular respiration reaction, these reactions are both very slow, and occur over the time period of billions of years. Weathering and metamorphism account for the movement of about 0.1 Gtons of carbon in the global carbon cycle .4
The third reaction present in the cycling of carbon is air water exchange. In this reaction, carbon dioxide gas dissolves at the surface of the atmosphere, and in the reverse direction, carbon dioxide gas is released from ocean into the atmosphere. The rate of the reaction is governed by differences in gas concentration in the ocean and the atmosphere and the exchange coefficient. The exchange coefficient determines how quickly a gas molecule can move from the ocean to the atmosphere and vice versa.
It takes hundreds of years for the carbon dioxide to reach equilibrium between the atmosphere and the ocean because of the amount of time that it takes for deep ocean water to reach the surface ocean, where it can exchange CO2 with the atmosphere. Once CO2 dissolves in the ocean, it undergoes a reaction to form carbonic acid, which then dissociates and increases the concentration of hydrogen ions in the water, making the water more acidic. This reaction allows the ocean water to act as a buffer, stabilizing the concentrations of CO2 in the water.
Carbon cycle - anthropogenic (fossil fuels)
The carbon in the atmosphere has increased by 133 ppm since 1750. Initially, the rise in carbon dioxide in the atmosphere was caused by land use changes and deforestation, which decreased the amount of photosynthesis that was removing carbon dioxide from the atmosphere. The clearing of land also took stored organic carbon and converted it to carbon dioxide, further increasing the CO2 in the atmosphere.
After the industrial revolution, the amount of carbon dioxide continued to increase due to the burning of fossil fuels. Fossil fuels include coal, oil, and natural gas.
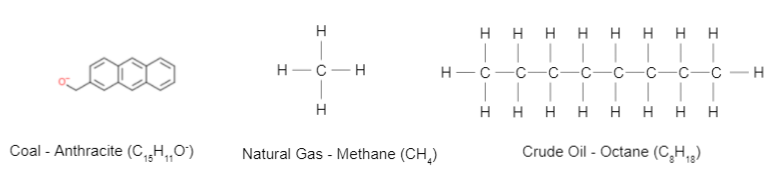
Figure 3: The chemical structures of coal, natural gas, and crude oil.
Coal is made up of organic carbon preserved in the land plants found in swamps and other organic rich soils and sediments, where the water keeps the carbon from becoming oxidized. The carbon accumulates into thick layers of peat. As the organic matter gets buried deeper in the ground, both the temperature and pressure increase. After a few million years under high temperature and high pressure, coal is formed. While the estimates of the amount of carbon contained in coal reserves differ, the low estimates assume that there are 1,000 Gtons of carbon currently stored in the coal reserves5. Oil is a hydrocarbon, made of hydrogen and carbon atoms. Petroleum (oil and natural gas) is mainly formed when dead plankton and algae sink to the ocean floor and mixes with the clay material that is deposited in the ocean by rivers and streams. If this occurs in an anoxic (without oxygen) environment, the organic material is converted into sedimentary rock called organic shale which is made of kerogen, which then continues to be buried in the Earth’s interior. The temperature and pressure increases, and the kerogen is converted into oil hydrocarbons, and at an even higher temperature, natural gas, mostly in the form of methane (CH4). Because the oil and natural gas are less dense than water, they migrate up towards the surface of the sedimentary rock where they are trapped by an impermeable layer of rock. This forms oil and natural gas reservoirs into which people can drill to release the oil and gas.
These materials are great sources of energy because they undergo combustion reactions, producing carbon dioxide, water, and large amounts of energy, in the reverse of the reaction described above. When one kilogram of coal is burned, it produces 25,000,000 joules (25 MJ) of energy. One kilogram of crude oil produces 42-47 MJ of energy. Natural gas has the highest energy production potential, producing 42-55 MJ of energy for every one kilogram of natural gas. Using the ratio of the molecular weight of carbon dioxide (MW= 44.01 g/mol) and the molecular weight of carbon (MW=12.01 g/mol), the amount of CO2 produced by the combustion of one kilogram of coal, oil, and natural gas can be calculated6. Anthracite coal is made of 90% carbon. The amount of carbon dioxide produced from one kilogram of coal is equal to (44.01/12.01)x 0.9 which equals 3.3 kg of CO2 released. Crude oil, or octane (C8H18) is composed of 84% carbon and therefore one kilogram of octane burned produces 3.1 kg of carbon dioxide. Methane (CH4), (natural gas) is composed of 75% carbon, so one kilogram of methane burned produces 2.75 kg of CO2.
These two major changes (deforestation and burning of fossil fuels) have significantly altered the carbon cycle, causing an imbalance. Deforestation increases the amount of CO2 in the atmosphere by decreasing the amount of photosynthesis occurring, as well as decomposing the organic carbon stored in plants and soils. Today, deforestation mostly occurs in the tropics. It is responsible for a 1.6 Gt increase of carbon per year. The burning of fossil fuels adds 9.4 Gt of carbon to the atmosphere per year. This leads to a total increase of 11.0 Gt of carbon.
Some of this carbon is removed by changes in the carbon sinks. Land uptake has increased by 3.4 GtC, having doubled over the last 50 years. There are several possible reasons for why land uptake has increased in the anthropogenic carbon cycle. One possibility is that with the warming atmosphere throughout the globe, the growing season has lengthened, which allows for a longer “inhale” in the breathing of the land. Another possible reason for the increase in carbon uptake on land is because of CO2 fertilization, which causes plants to grow faster. This occurs because when there is an increase of carbon dioxide in the atmosphere, the amount of time that the plant stomata has to be open decreases, which leads to higher rates of photosynthesis. It remains, however, unclear how much of an effect this will have on future carbon levels7. As the world continues to warm, it is possible that the increasing temperature of the soil will inhibit the soil from storing carbon, therefore decreasing the amount of carbon that is held in the land reservoir.
In the anthropogenic era, carbon uptake by the ocean has increased by 2.5 GtC. This is mainly due to the equilibrium shift in the air water exchange reaction. When the concentration of carbon dioxide in the water increases, the reaction is shifted to the right, consuming more CO2, and producing more HCO3-. This reaction acts as a buffer for the water, and allows the seawater to take up a large amount of carbon dioxide. The amount of CO2 being taken up by the ocean has also increased due to the reversed concentration gradient where the concentration of carbon dioxide in the atmosphere is much larger than the concentration of carbon dioxide in the ocean. Carbon dioxide, therefore, moves into the ocean through diffusion in order to try and reach a new equilibrium state.

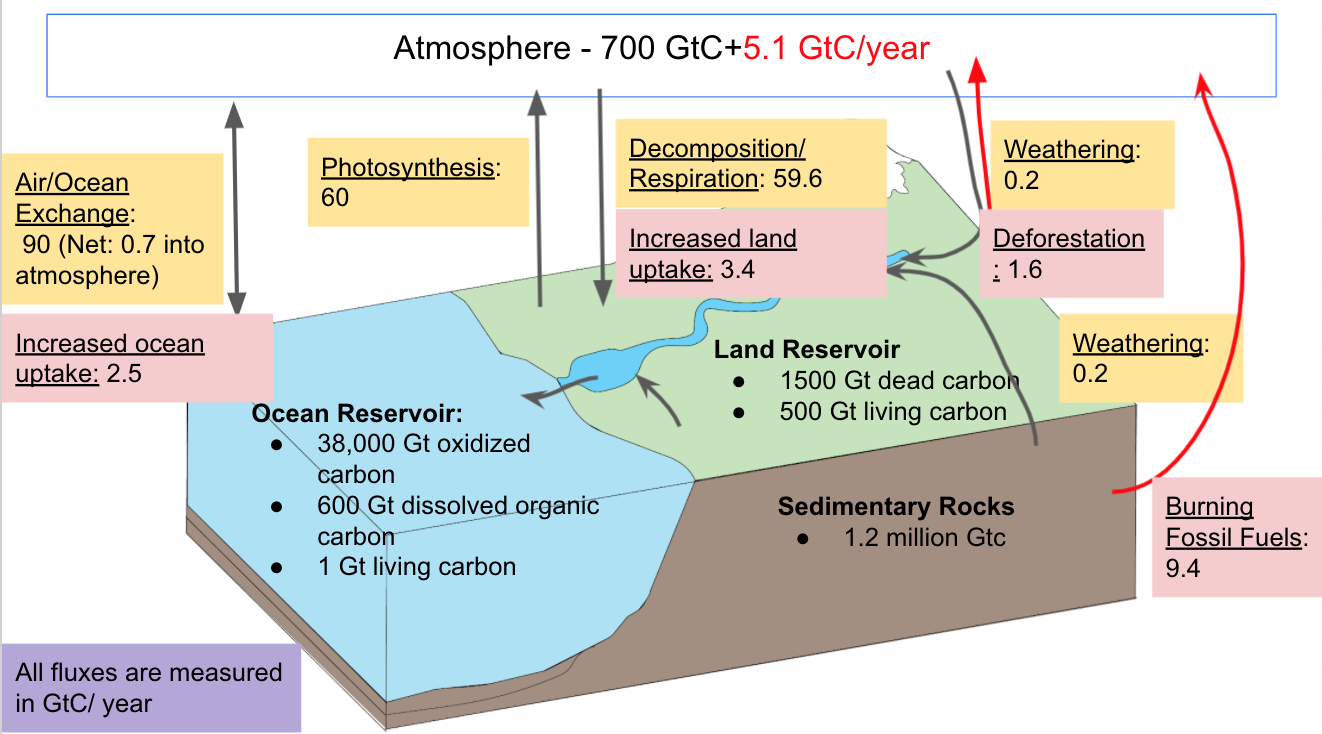
Figure 4: The anthropogenic carbon cycle includes changes in the carbon reservoirs due to the burning of fossil fuels and land use changes
The total increase in carbon in the atmosphere (11.0 Gt) is partially balanced by the 3.4 Gt decrease due to land sinks and 2.5 Gt decrease due to ocean sink, for a total decrease of 5.9 Gt. This leads to an overall increase of 5.1 Gt of carbon dioxide in the atmosphere8. This can be represented by the equation for the global carbon budget: Ef+Eluc=Gatm+Socean+Ssland+Bim where Ef is fossil fuel emissions, Eluc is the land use changes, Gatm is the carbon growth in the atmosphere, Socean is the carbon stored in the atmosphere, Slandis the carbon stored on land, and Bim is equal to the carbon imbalance.
How carbon dioxide affects temperature
As can be seen in the graph below, the global temperature has steadily increased throughout the last century.
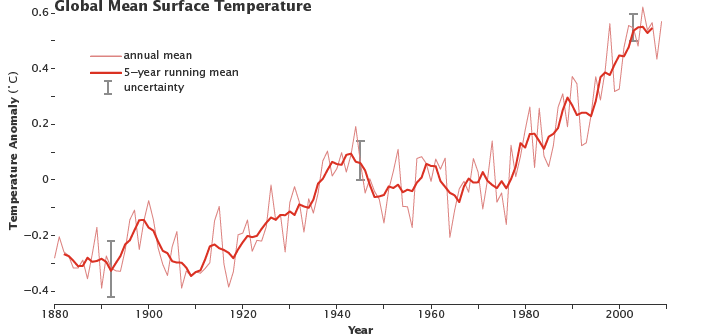
Figure 5: Figure 4: This graph shows the temperature anomaly from 1880 to the present. In 2010, the 5 year mean is 0.5 degrees celsius, meaning that the average temperature is 0.5 degrees higher than the average temperature. (Source: NASA)
The main cause of this increase is due to increased greenhouse gases in the atmosphere. The dominant greenhouse gases are carbon dioxide, water vapor, methane, nitrous oxide, and hydrofluorocarbons. Between 1990 and 2015, it is estimated that greenhouse gas emissions have increased by 43%, with more than 50% of the greenhouse gases emissions coming from carbon dioxide. The amount of carbon dioxide in the atmosphere is currently 409.8 ppm, which is 100 ppm higher than the highest point it has been in the last 800,000 years.
The Greenhouse effect is the process by which greenhouse gases trap heat in the atmosphere. Without the greenhouse effect, the temperature of the Earth would be too low to be habitable. The implications of the greenhouse effect can be seen when comparing Earth to other planets. Mars, which has very low CO2 in the atmosphere, has an average temperature of -50 ℃. On the other hand, Venus, with its atmosphere made up of 96 % CO2, has an average temperature of 420℃. Earth is found in the middle of these extremes, with an atmosphere that contains 0.03% carbon dioxide and an average temperature of 15℃.
Greenhouse gases absorb and emit thermal radiation from the sun. Much of the radiation emitted from the sun is short wavelength radiation which includes visible light and portions of infrared (IR) radiation. When IR radiation hits a CO2 molecule, it causes the bonds to vibrate. The CO2 molecule can then emit the extra energy as an infrared photon or transfer it to another molecule when they collide. This causes the motion of the surrounding molecules to increase, which increases the average kinetic energy, or temperature of the molecules. When this process occurs naturally (without the human added greenhouse gas emissions), the radiation entering the atmosphere is equal to the radiation emitted back out into space. This is called the equilibrium climate.
When the amount of carbon dioxide in the atmosphere increases, more heat is trapped, disrupting the climate equilibrium. Radiative forcing is a measure of how much the Earth’s energy system is out of balance, measured in Watts per meter squared (W/m2). It can be found by measuring the amount of energy going in and subtracting the amount of energy reflecting back into space, and usually considers the energy at the top of the atmosphere, at the boundary between the troposphere and stratosphere. The current level of radiative forcing is 2.3 W/m2 from 1750 to 2011 according to the IPCC (Intergovernmental Panel on Climate Change) report. This is 43% higher in 2011 than it was in 2005. Carbon dioxide accounts for the largest percentage of the radiative forcing, when compared to other greenhouse gases. While carbon dioxide absorbs less heat than methane or nitrous oxide, it has a much higher concentration in the atmosphere and stays in the atmosphere for a longer period of time.
Climate sensitivity models
Climate sensitivity is a measure of how much the Earth’s temperature will change due to the human caused increase in carbon dioxide. Because the climate system is a complicated system with many different inputs and feedback cycles, there is not an easy answer to this question - scientists have come up with many different models to explain how the rise in carbon dioxide will affect the Earth’s temperature. In a simple model that does not include any feedback systems, the temperature is predicted to increase by one degree Celsius when the concentration of carbon dioxide in the atmosphere doubles. However, there are several feedbacks in the climate system that alter this prediction. Positive feedback amplifies the effects warming to further increase the temperature, and negative feedback diminishes the effect of warming. Some of the main feedbacks that affect the climate sensitivity are water vapor concentrations, clouds, and surface albedo (reflectiveness of the earth’s surface).
Air can hold more water vapor when warmed. Therefore, when the temperature increases, the amount of water vapor in the atmosphere increases. This, in turn, increases the greenhouse effect by trapping more heat. The effect of clouds on warming is more complicated. Clouds at low altitude reflect some sunlight back into space and decrease the temperature. High altitude clouds trap more heat in the atmosphere. Additionally, clouds that have more liquid water molecules are more effective at blocking sunlight than clouds composed of ice. The third main input is albedo, the measure of reflectiveness of the Sun on the Earth. As snow and ice cover decrease with increasing temperature, the albedo will also decrease because there is less reflective material. Given these inputs, estimates of climate sensitivity range from an increase of 1.5 degrees Celsius to 4.5 degrees Celsius when the carbon dioxide in the atmosphere doubles, with an average estimate of 3 degrees.
There are several different methods used to estimate climate sensitivity, all of which provide different estimates as to how much the temperature will increase when carbon dioxide doubles. The physics based method uses fundamental physics equations and models to predict how the temperature will increase. For example, the first law of thermodynamics, which describes the flow of energy in the Earth’s system and states that energy cannot be created or destroyed, only transformed. Another example is the Clasius Clapeyron Equation which describes the relationship between temperature and water vapor pressure. These (and other) classical physics equations are imputed into a model, which then predicts the temperature increase if the carbon dioxide is doubled. A second method to estimate climate sensitivity uses direct observations and records of surface temperature and ocean heat. This method typically yields lower temperatures than the other methods. This is partially because of uncertainty in the heat of the ocean due to incomplete records, especially in places like the Artic where the temperature is rapidly increasing. The lower estimates can also be accounted for by the fact that these estimates do not take into account the effect that modern feedback loops play in the changing temperature9. These estimates are also affected because the role of aerosols in warming is not yet fully understood. Some aerosols lead to cooling while others lead to warming, and the aerosols also interact with clouds in a way that may increase cooling. A third method of estimating climate sensitivity is by looking at paleoclimate changes. In order to do this, scientists look at fossils and other natural records such as tree rings in order to learn about “climate proxies” from thousands to millions of years ago. Through this method, researchers can learn about how the climate has changed in different temperature periods. Because of the different time periods stressed in each of these models, the different models yield slightly different results for the climate sensitivity of the Earth. They all, however, agree that the Earth is warming at a dangerous rate and will continue to warm unless drastic actions are taken.