Walt Whitman said:
-
"I bequeathe myself to the dirt, to grow from the grass I love;
-
If you want me again, look for me under your boot-soles."
Ecosystems and the cycles of nutrients are not as simple as Whitman describes in the above quote, however, it gives the reader an image of living things undergoing changes. Scientists describe these changes as Nutrient Cycles.
The elements that are essential for living organisms: oxygen, carbon, nitrogen, phosphorus and sulfur are generally stable and remain preserved in the ecosystems of Earth undergoing changes called cycles. Water, although it is not an element, is essential for all known life, and also is mostly preserved through cycles. The cycles of water and the aforementioned elements can involve the simple process of the substance changing states and locations, or they can involve a series of chemical changes in which they are integrated into different molecular compounds. Microbes play important roles in the functioning of these cycles often as primary producers through photosynthesis and respiration or as detritivores through fermentation and respiration, and through other chemical processes (Sengbusch 2010).
Water Cycle
The amount of water on the earth is finite. Water can be cycled through different states and through ecosystems but it cannot be reproduced. The Water Cycle includes the processes of evaporation, transpiration, condensation, precipitation and collection. During evaporation and transpiration liquid water is changed into water vapor. Condensation occurs when water vapor returns to a liquid state. Precipitation happens when the condensed water droplets or ice crystals fall to earth. Collection refers to the areas on earth where water collects such as ponds, lakes oceans and streams. (Kidzone 2010).
WATER CYCLE DIAGRAM
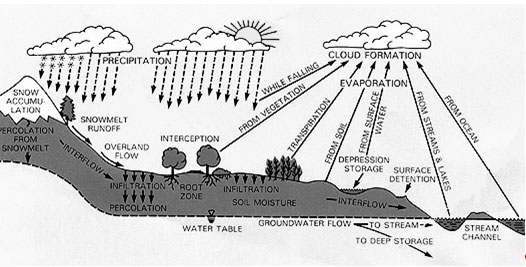
http://rst.gsfc.nasa.gov/Sect16/Sect16_4.html
Oxygen Cycle
Plants and photosynthetic bacteria play an essential role in the oxygen cycle primarily through the process of photosynthesis. Only one two-thousandth of the entire earth's atmospheric oxygen is produced by plants each year the remainder comes from photosynthetic bacteria. In turn, animals and other living things use that oxygen for essential life processes and release other gases, mainly CO
2
back into the atmosphere which plants use again for photosynthesis. (Sengbusch 2010).
Earth's early atmosphere, however, was anoxic or without oxygen. Gases in Earth's early atmosphere included: carbon dioxide, nitrogen, ammonia, and methane. In the early atmosphere of Earth, the type of photosynthesis that we are familiar with, which produces oxygen, did not occur. There was another type that still occurs today all around us. It is a much simpler process and is known as anoxygenic photosynthesis because it does not produce O
2
. This takes place in green and purple sulfur bacteria, which use H
2
S, which is produced by sulfate-reducing bacteria mentioned above, in place of H
2
O used by oxygen-producing photosyntesizers like green plants and algae. These two types of bacteria are important drivers of Earth's Sulfur Cycle. In both types of photosynthesis, the organism uses light to make ATP, the main energy carrier of cells (Madigan, and Martinko 29, 129, 302).
Carbon Cycle
The carbon makes up about 50% of the biomass of each living organism. It is also contained in earth's atmosphere, the ocean, rocks and soil. Carbon and oxygen atoms are combined as carbon dioxide (CO
2
) molecules in the air. CO
2
is used by autotrophs, including plants and photosynthetic bacteria primarily in the Calvin Cycle for CO
2
fixation. CO
2
is not used in the light reactions of photosynthesis, it is used in the Calvin Cycle which converts inorganic carbon in the form of CO
2
into organic carbon or biomass that makes up the tissues of the plant. Then, it is available as food for organisms higher on the food chain. When plants die they may become buried and become fossil fuels after many millions of years. When these fuels are burned carbon dioxide returns to the air. (UCAR 2010).
CARBON CYCLE DIAGRAM
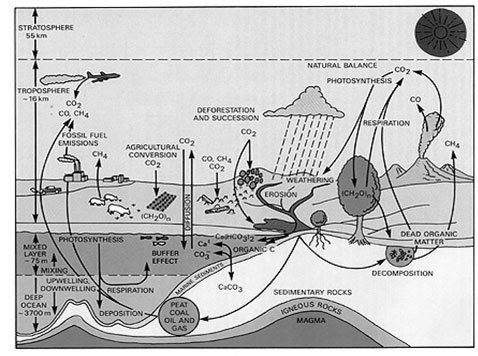
http://rst.gsfc.nasa.gov/Sect16/Sect16_4.html
Nitrogen Cycle
Earth's atmosphere is nearly 80% Nitrogen (N
2
). Organisms need it to produce proteins and DNA. However, living things cannot make use of nitrogen gas directly from the atmosphere. Microbes in the soil, called diazotrophs and aquatic microbes like cyanobacteria are essential for changing or "fixing" atmospheric nitrogen into ammonium (NH
4
+
), which is the form that plants use to make proteins. Some plants, mainly legumes, have nitrogen-fixing bacteria in their root nodules, also clovers, which we will explore in a hands-on exercise. Once nitrogen is in a usable form in plants, other organisms that eat plants also have access to nitrogen. After plants, animals and other organisms die, microbes and other decomposers feed on them changing their proteins back into nitrogen gas that is released into the atmosphere. The formula for nitrogen fixation is: N
2
+ 8H→ 2NH
3
+ H
2
. (Madigan, and Martinko, 641), (Kids Know it Network 2010).
NITROGEN CYCLE DIAGRAM
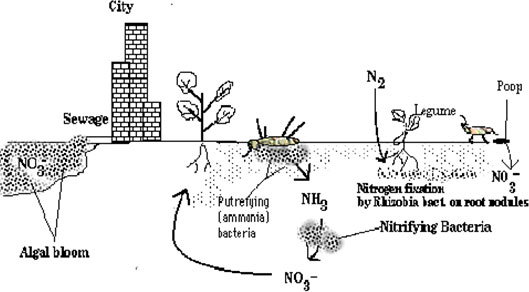
http://soil.gsfc.nasa.gov/NFTG/nitrocyc.htm