Stephen C. Kissel
Matter Defined
Matter is any substance that has mass and volume. If it can be proven that a substance has mass and takes up space, it is matter. Energy is the capacity to cause change and is not matter. Light energy, sound energy, heat energy, potential energy, and kinetic energy are examples.
Mass is the amount of matter. For instance, a cake has more matter that a slice of that same cake. It can be measured in milligrams, grams, or kilograms using an analytical electronic balance or a triple beam balance. This is different than using a scale to determine weight because the reading on a scale for a particular object depends on the force of gravity. Gravity is different on different celestial bodies, so weight changes according to where it is measured. Mass does not take into consideration gravity and is constant. Furthermore, mass has a positive correlation with inertia. The more massive an object is, the more inertia it has. An object with a lot of mass is more difficult to put in motion or stop if already in motion compared to an object with a small amount of mass.
Volume is the amount of space taken up. For instance, a volley ball has a larger volume than a golf ball because it takes up more space. Volume is three-dimensional and incorporates length, width, and height. It can be measured in cubic millimeters or cubic centimeters. Volume can be measured using a meter stick to measure the sides of a regularly-shaped object. To measure an irregularly-shaped object, water displacement can be determined using, for example, a graduated cylinder (assuming the object has a higher density than water). The amount of water in a graduated cylinder can be noted before an object is placed in it and after the object is placed in it. The difference of these measurements is the volume of the object.
Basic Unit of Matter
https://www.thinglink.com/scene/719221188345200641
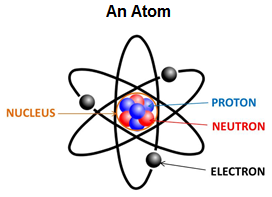
An atom is the basic unit of matter. Is the smallest part of a chemical element that retains the properties of that element. An element is matter that is made up of one type of atom. There are over 110 chemical elements. Since there are over 110 elements, there are over 110 types of atoms. Everything that you see, smell, hear, taste, and touch consists of atoms.
Atoms are made up of three sub-atomic particles called protons, neutrons, and electrons. Protons are positively charged and located in the nucleus, the center of the atom. The number of protons determines which element the atom represents. Each element has a different number of protons. The number of protons is the element’s atomic number. Neutrons have no charge and are also located in the nucleus. The nucleus makes up more than 99.9% of an atom’s mass. Electrons are the smallest of these particles and travel in shells or orbitals around the nucleus, sometimes called the electron cloud. If the number of protons equals the number of electrons, the atom has no charge. If there are more protons than electrons (or vice versa) it is an ion. Electrons allow atoms to bond with each other and form molecules. A molecule is two or more atoms bonded together. When a metal element loses electrons becoming a positively charged cation and a non-metal element gains electrons becoming a negatively charged anion, an ionic bond is formed. It is valance electrons, or outermost electrons, that are transferred when ionic bonds are formed. When non-metal elements share pairs of electrons with other non-metal elements covalent bonds are formed.
|
|
Subatomic Particles
|
|
Proton
|
Neutron
|
Electron
|
|
Symbol
|
p or p
+
|
n or n
o
|
e or e
-
|
|
Charge
|
+1
|
0
|
-1
|
|
Mass
|
1 amu
|
1 amu
|
.00054 amu
|
Classification of Matter
Matter is either a pure substance or a mixture. A pure substance cannot be taken apart using physical means. Elements are pure substances made of one type of atom. An example is aluminum. Compounds are pure substances made of atoms from two or more elements. Water, or H
2
O, is an example. A mixture is two or more pure substances in varying amounts that can be taken apart by physical means. Air would be an example since it is primarily a mixture of N
2
and O
2
gas. Homogeneous mixtures, also called solutions, are mixed uniformly. Sugar dissolved in water is an example. Heterogeneous mixtures, also called suspensions, are not mixed uniformly. An example is concrete. Suspensions settle out over time, like corn starch and water. Colloidal suspensions, or colloids, do not settle out over time, like milk.
Properties of Matter
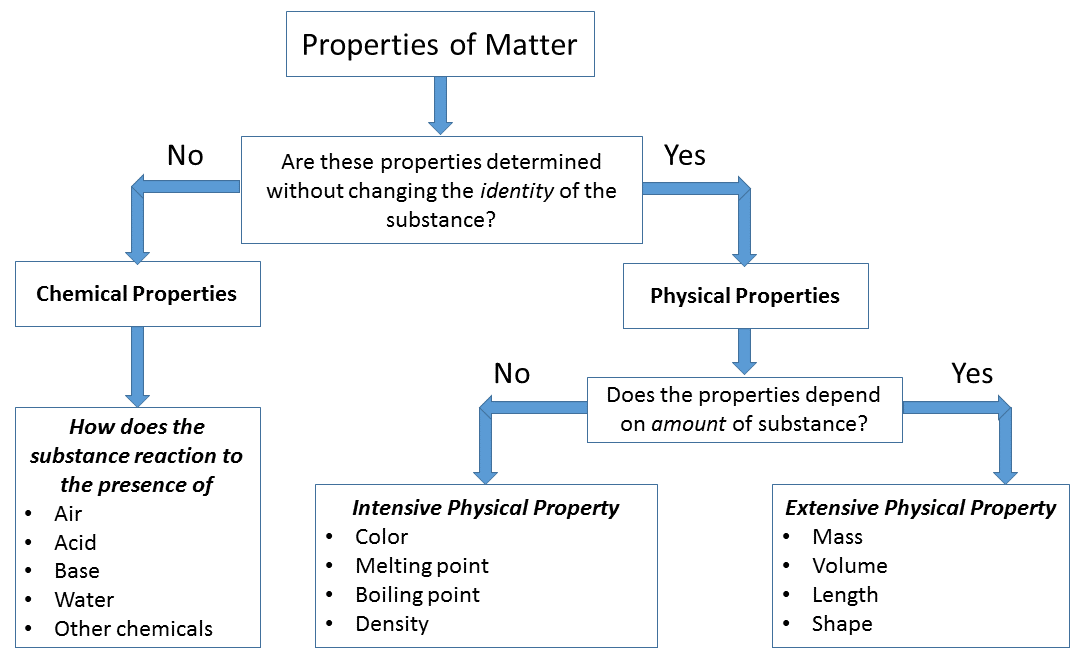
A property is a special or distinctive attribute, quality, or characteristic. Properties can be used to identify matter. For example, different types of matter can be described by electrical conductivity, thermal conductivity, color, melting point, mass, density, or volume. Therefore, these are properties of matter.
http://chemwiki.ucdavis.edu/Core/Analytical_Chemistry/Chemical_Reactions/Properties_of_Matter
Chemical properties are observed when one substance reacts with another substance and changes into a different substance. For example, when iron reacts with oxygen, it rusts and becomes reddish-brown flakey iron oxide. The ability to rust is a chemical property. Furthermore, when fire burns beige hard wood it turns into black powdery ash. This chemical property is flammability. Other chemical properties include reactivity with acids, bases, and water.
Physical properties are observed without a substance changing into a different substance. Generally, you can detect them with your senses. There are intensive and extensive physical properties. Intensive physical properties do not depend on the amount of matter present. They include color, melting point, malleability, ductility, thermal conductivity, electrical conductivity, ability to dissolve other substances, and density. For example, the density of half a cube of lead is the same as the density of the cube of lead before it was cut in two. It is not dependent on the amount of lead present. Extensive physical properties depend on the amount of matter present. Mass, volume, and length are typical examples. The mass of half a cube of lead is half of the mass of the cube before it was cut in two, and the volume of a piece of steel depends on the amount of steel present.
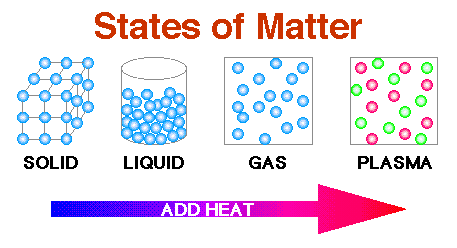
States of Matter
A state (or phase) is the way matter organizes its atoms and molecules and is a physical property of matter. Common states of matter include solids, liquids, and gases. Plasmas are a less common state of matter.
http://questgarden.com/75/22/0/081214000133/task.htm
A solid has a definite shape and does not conform to the shape of a container. It has a definite volume, as well. The particles of a solid are arranged in an organized pattern and are tightly packed such that they move very little and are not easily compressed. They can vibrate around their equilibrium position, but their atoms are in a fixed position and do not move throughout the solid. Solids can be crystalline or amorphous. The particles of crystalline solids are arranged in an organized repeating pattern. Examples include diamond, graphite, and table salt. When broken, they have straight edges. They also have exact melting points. The particles of amorphous solids have no regular pattern. Examples include glass, plastic, and rubber. When broken, they have curved edges. They do not have exact melting points, so they become soft and gooey before turning into a liquid.
A liquid has particles that flow past each other, so it has an indefinite shape and takes the shape of the container in which it is poured. The particles of a liquid, which have greater kinetic energy than a solid, are still relatively close to each other so it has a definite volume. A liquid is very difficult to compress and has a top surface. A liquid has surface tension because like particles have cohesion and are attracted to each other more than to other particles with which they may be in contact. Surface tension allows an insect like a water strider and a reptile like a basilisk lizard to “walk on water.” Cohesion is stronger below the surface of a liquid where like particles are surrounded by each other. This causes a liquid to form a sphere. When gravity acts on a drop of rain, small raindrops stay spherical, medium-sized ones flatten a bit, and large ones become teardrop-shaped. The particles of a liquid are also attracted to different particles, like those of a container, because of adhesion. Acting together, cohesion and adhesion create a meniscus or concave shape at the surface. If a liquid is in a graduated cylinder, the mark at the base of the meniscus should be used to measure the volume. Adhesion is also the reason for capillary action, the movement of a liquid up a narrow tube. Water drawn up the xylem of a bonsai tree or a giant sequoia tree are examples of this. The term viscosity refers to how freely a liquid flows. For example, maple syrup is more viscous, or thicker, than water.
A gas has enough kinetic energy (thermal energy) to overcome intermolecular forces and spread apart, so it has an indefinite shape and volume. Without the confines of a container, a gas will spread without limit. If in a container, a gas will take on the shape and volume of the container. Since gas molecules are far apart from each other, a gas can easily be compressed. The volume, temperature, and pressure of a gas are interrelated. Boyle’s law states that gas particles will collide more often in a container with less volume, thus creating more pressure if temperature is held constant. Charles’ law states that gas particles will increase their movement and occupy more volume when temperature is raised if pressure is held constant. To maintain that pressure, the volume of a container must increase when a gas is heated. Avogadro’s law states that gases contain an equal number of molecules when the temperature, pressure, and volume are the same. The ideal gas law is a combination of these laws and is written PV = nRT, where P, V, and T are pressure, volume, and temperature, respectively, n is the number of moles (or essentially, the amount of substance), and R is a constant. Kinetic Molecular Theory explains the behavior of gases. It is based on the following assumptions:
-
Gases are composed of particles in constant motion.
-
The particles move in straight lines unless they collide with each other or the wall of their container.
-
The particles are much smaller than the space between them, so a gas is mostly empty space.
-
Collisions are elastic and no energy is lost.
-
The average kinetic energy of the particles depends on the temperature of the gas.
A plasma is a charged gas of particles with extremely high kinetic energy. Therefore, a plasma has an indefinite shape and volume. Particles are very far apart. Some plasma atoms lose electrons making them into positively charged ions. Stars, with extremely high temperatures, are plentiful and made of plasma which means that plasma is the most abundant state of matter in the universe. However, on earth, they are less common than other states of matter, but examples exist such as neon lights and the aurora borealis.
|
|
States of Matter
|
|
Solid
|
Liquid
|
Gas
|
|
Shape
|
Definite
|
Takes
shape of
container
|
Takes
shape of
container
|
|
Volume
|
Definite
|
Definite
|
Takes
volume of
container
|
|
Particle Position
|
Tightly packed
|
Less
closely
packed
|
Very far apart
|
|
Particle Motion
|
Very little
motion,
Vibrate within limited area
|
Moderate
motion,
Flow around each other
|
Constant
random motion,
Fly in all directions at great speeds
|
|
Compressibility
|
Not easy
to
compress
|
Not easy
to
compress
|
Easy to compress
|
All matter has thermal energy, or heat. Heat is the total energy obtained from the movement of particles. A greater amount of particle movement means more heat. For instance, the human body shivers when it is cold to generate more heat for itself. Heat flows from a warmer object to an object that is cooler. Temperature is a measure of the average kinetic energy of particles. If a pail has just been dipped into the ocean, the water inside the pail is the same temperature as the ocean. The ocean, though, has much more heat content as it is vast in comparison to the pail of water.
Heat can be transferred in three ways. Convection is the transfer of heat through a fluid, namely a liquid or a gas. As the fluid heats, its particles move farther apart and rise. Cooler particles move in below, thereby creating circular convection currents. Water being heated in a pot on the stove moving in a circular pattern is an example. Conduction is the transfer of heat through a solid. Heat from a source on one end of a solid making particles move more will travel along the solid and make adjacent particles move more. Heat traveling up the handle of a metal spoon partially submerged in boiling water is an example. Radiation is the movement of electromagnetic waves and can occur without a medium. Sunlight that travels to Earth through space is an example.
Density is mass per unit volume, another physical property of matter. Density varies with pressure and temperature, affecting gases more than solids or liquids. An increase in pressure decreases volume and therefore increases density. An increase in temperature generally increases volume and therefore decreases density. Different elements have different densities. Sometimes an element can even be identified by its density. Density is related to buoyancy and purity.
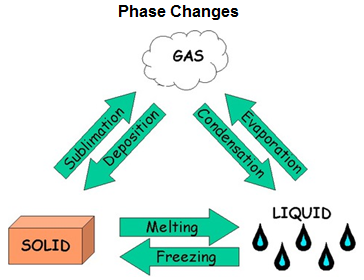
Phase Changes
Phase changes occur when a substance transforms from a solid, liquid, or gas into a different phase or state. All substances change phases depending upon temperature and pressure. Each substance has unique intermolecular forces and these influence the temperature and pressure at which substances will change their state. When a substance does change, two phases exist at the same time as the transition occurs. There are six categories of phase change, each being reversible:
-
Melting or fusion is when a solid turns into a liquid.
-
Freezing or solidifying is when a liquid turns into a solid.
-
Evaporation or vaporization is when a liquid turns into a gas.
-
Condensation is when a gas turns into a liquid.
-
Sublimation is when a solid turns directly into a gas.
-
Deposition is when a gas turns directly into a solid.
"Quizlet." Chemistry. Accessed June 28, 2016. https://quizlet.com/80471746/chemistry-phases-flash-cards/.
Melting, evaporation, and sublimation are endothermic processes. The substances absorb energy from the surroundings which enables the change to occur. A way to remember this is to think of “endo” as entering. Energy goes into, or enters, the substance. Therefore, the surroundings are cooled.
Freezing, condensation, and deposition are considered exothermic. The substances release energy into the surroundings enabling the change to occur. A way to remember this is to think of “exo” as exiting. Energy goes out of, or exits, the substance. Therefore, the surroundings are heated.
|
Phase Changes
|
|
|
Phase to Phase
Change
|
Endothermic or Exothermic?
|
Example
|
|
Melting
|
Solid to Liquid
|
Endothermic
|
Ice Cube to
Water
|
|
Freezing
|
Liquid to Solid
|
Exothermic
|
Sweetened Cream to Ice Cream
|
|
Evaporation
|
Liquid to Gas
|
Endothermic
|
Puddle to Water Vapor
|
|
Condensation
|
Gas to Liquid
|
Exothermic
|
Water Vapor to Dew Drops
|
|
Sublimation
|
Solid to Gas
|
Endothermic
|
Dry Ice to Carbon Dioxide Gas
|
|
Deposition
|
Gas to Solid
|
Exothermic
|
Water Vapor to Frost
|
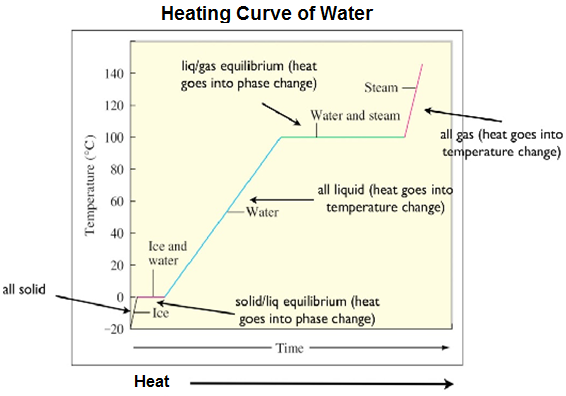
http://ch301.cm.utexas.edu/section2.php?target=thermo%2Fenthalpy%2Fheat- curves.html
Temperature can enable a phase change to occur. Heating an ice cube, a solid, over a hot plate changes it into water, a liquid. The substance changes when its melting point is reached. Melting point is the temperature at which a solid turns into a liquid. With continued heating, the water, a liquid, will change into water vapor or steam, a gas. The substance changes when its boiling point is reached. Boiling point is the temperature at which a liquid turns into a gas.
Pressure can also enable a phase change to occur. An increase in pressure, at a constant temperature, can cause a gas to turn into a liquid and a liquid to turn into a solid. The reverse can happen with a decrease in pressure.
When a substance is heated two things can happen. The temperature of the substance can increase or a phase change can occur. If heat is added to a solid it will get hotter until its melting point is reached. At that point, the added heat will enable a phase change from a solid to a liquid, but the temperature will not rise. Then, once all of the solid melts, the liquid will get hotter as heat continues to be added. When the boiling point is reached, a phase change will occur and the liquid will turn into a gas. The plotting of temperature versus time is called a heating curve. It is level during phase changes because the added heat is used to facilitate the change and break intermolecular forces rather than increase the temperature.