Collision Theory Lab
Purpose
Students will explore the principles of the collision theory.
Materials
Soft-tipped darts, Balloons, Tape
Preparation
The classroom will be cleared of all chairs. Inflated balloons will be taped to an appropriate wall or backing. A line ten feet behind the wall of balloons will be marked with tape.
Procedure
-
Five students at a time will be asked to step up to the line and throw plastic darts at the balloons.
-
Students will find that as they throw darts some will hit the balloons dead on, but not with enough energy. Other times, a student will throw a dart and it will have enough speed, but hit the balloon off-angle. Only those darts thrown with the correct orientation (towards the center of the balloon) and enough energy (enough speed of the dart) will result in a popping of the balloon or creation of the product, in this case.
-
Students that are not throwing darts will take notes on the collisions and fill in the following table. In addition, students will be asked to track three collisions that result in a popped balloon and three collisions that do not end up in a popped balloon and make notes as to why each instance resulted the way it did.
|
Relative Speed of Dart
|
Relatively slow speed
|
Medium speed
|
Relatively fast speed
|
|
Resulting Interaction
|
|
|
|
-
Students will rotate from being observers to participants and get a chance to throw darts at the balloons.
Questions
-
In this particular simulation, what do you think was more important in getting the balloon to pop, orientation or energy? Explain with evidence.
_______________________________________________________________
-
How do you think this actually relates to the way in which reactions occur?
_______________________________________________________________
Michaelis-Menten Lab
Purpose
To understand how saturation occurs according to Michaelis-Menten kinetics and why K
m
is important.
Materials and Methods
Two containers, Popsicle sticks, Blindfold, Timer
Explanation
The student volunteer is the enzyme. The hand of the student grasps the Popsicle stick and represents the enzyme’s active site. The catalytic event is the breaking of the Popsicle stick, which will follow a transfer from the left container (containing the reactants, or Popsicle sticks) to the right container (containing the products, or broken sticks).
Procedure
-
Popsicle sticks will be put in the left container and randomized for each trial with the number of Popsicle sticks from 5 – 40 to simulate different “concentrations.”
-
Then, a student is blindfolded and is asked to break Popsicle sticks for ten seconds, placing the pieces into the right container. This is done to get the initial rate of the reaction.
-
Then the number of Popsicle sticks is varied and the exercise is repeated.
-
Students will create a graph depicting V
0
as a function of substrate concentration to get the saturation curve.
Results
Once the data and necessary information is collected a graph similar to Figure 4 will be created.
This graph will show that the rate of Popsicle stick breaking (V
0
) depends on the Popsicle stick concentration. Additionally, it will show that as Popsicle stick concentration is increased, saturation occurs and the rate of the reaction reaches some maximum (V
max
).
Exploring
K
m
To determine the point at which this occurs requires manipulation of the Michaelis-Menten equation. If you set, [S] = K
m
in the below equation and simplify, you get the equation just below that one, which shows that, indeed, K
m
is the concentration of the substrate when V
0
is half of V
max
.
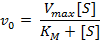
V
0
= V
max
/2
Using the saturation curve, students will determine the K
m
for the reaction.
Why is K
m
important?
K
m
serves as a dissociation constant such that larger K
m
values mean lower affinity between the substrate and enzyme and smaller K
m
values mean the higher affinity between the substrate and the enzyme.
Questions
1. Why does the reaction reach a saturation point?
_______________________________________________
2. Repeat this activity with other people acting as enzymes and breaking Popsicle sticks. Generate saturations curves for each enzyme and determine the respective K
m
values. Which enzyme had the highest affinity for the substrate? Which enzyme had the lowest affinity for the substrate?
_______________________________________________
3. How did low or high K
m
values correlate to what you saw when people were breaking Popsicle sticks?
_______________________________________________