Overview
This unit focuses on thermodynamics, and can be placed early in the curriculum, following introductory units on inquiry skills and describing matter. As one of the first content-specific units in chemistry, this unit lays a strong foundation in understanding the underlying principles of physical chemistry. Students will develop skills in inquiry and data to discover scientific knowledge by basing their conclusions on the data, and also recreating the laws of thermodynamics. Students will use this knowledge to model physical change during phase transitions and chemical changes during chemical reactions. The entire unit will have an overarching application that directly applies to the community, with each lesson addressing an aspect of the issue. As a foundationary unit, I expect to refer back to the skills from this unit in future units, particularly in the kinetics and equilibrium units.
This unit walks students through the laws of thermodynamics and energy, and then the manner in which these laws apply to chemical and physical changes. First, students will explore the concepts of energy, entropy, and enthalpy in closed systems. This will include discussions of the conservation of energy, and the differences between heat and temperature. Then students will apply these concepts to physical changes, including how changes to energy, enthalpy, and entropy changes cause and can be quantified during a phase change. Students will then cover chemical change and the prerequisites required for chemical change to occur. Each essential question below requires a certain number of lessons to focus on content. However, it should be noted that some lessons may require additional practice in math content, whether they are added on to focus on an individual concept, or the math concepts are rolled into the following lessons depends on the preference of the teacher. Lessons are recommended to spend 45 minutes focusing on content exploration and explanation, and 45 minutes focusing on expanding and practicing skills—either split into two class periods, or as one block. This unit should take approximately 4 weeks: 1 introductory lesson, 7-9 content lessons, and 2 wrap up lessons.
Energy and the Laws of Thermodynamics
1.
What Is Energy, and Can It be Created or Destroyed?
(1 Lesson)
Colloquially, the word energy is used to describe a person’s level of enthusiasm, or ability to complete a task. Not too differently, energy is defined in science as “the capacity to do work or to produce heat” (Zumdahl & Zuhmdahl, 2010). Energy can be manifested in many forms, but typically within two branches: Potential and kinetic energy, or alternatively, given off as heat. Potential energy is energy that is stored, whether by position or by composition. For example, a ball at the top of a very tall hill possesses a lot of potential energy relative to an identical one at the bottom of the hill due to its height (and the fact that gravity is pulling downward). Similarly, a compound with a complex structure holds a great deal of potential energy in its chemical bonds. This stored energy, whether due to composition or position, can be converted into kinetic energy, or energy due to motion, but also dependent on the mass. For example, the ball’s potential energy at the top of the hill is converted into kinetic energy once it starts rolling down the hill. Kinetic energy can be converted back into potential energy, such as when a ball rolls up a hill and then stops.
While energy can interchange between forms, the total amount of energy stays constant. The law of conservation of energy states that “energy can be converted from one form to another but can be neither created nor destroyed” (Zumdahl & Zuhmdahl, 2010). To be clear, the laws of thermodynamics do not consider nuclear reactions, wherein mass can be converted into energy. This law is also considered the first of three laws of thermodynamics. Energy can be converted from one type into another, for example, the chemical energy stored in the bonds of a compound can be released in an explosion, causing motion, radiant energy of electromagnetic waves, and sound energy. Transformation of energy is typically seen in machines, converting from potential to kinetic or heat, such as in a flashlight, or a car. Stereotypical Rube Goldberg machines involve a series of complex energy transformations within a series of reactions or events that culminate in the performance of a simple task.
The internal energy (E) of a system is made up of the sum of kinetic (KE) and potential (PE) energy of its components (both of which have units of Joules). The first law of thermodynamics states that energy of the universe is constant, although energy can change forms through either work (w)
or
heat (q), which also have units of Joules. Thus, the change total internal energy of a system can be represented by the equation:
∆E = q + w
In other words, the change in internal energy of the system is equal to the sum of the work done on it (compressing a gas, for example), and heat that flows into it. Work is typically seen as kinetic energy, such as expansion or compression of a piston (Zumdahl & Zuhmdahl, 2010). It is not necessary for students to be able to calculate the total internal energy of a system at this stage, but rather to understand the concept that energy is transferred as both work, in the form of kinetic energy, and heat.
2.
What is Heat, and How Does It Relate to Enthalpy and Temperature?
(2 Lessons)
Sometime energy is not perfectly converted from one form into another via work, but instead transfers energy into a secondary form, heat. Heat is the transfer of energy due to a difference in temperature, where temperature is defined as the movement of particles (Zumdahl & Zuhmdahl, 2010). In a “hot” object, the particles have large velocities, on average, and in a “cold” object, particles do not move as rapidly. Heat flows from a hot source, where the kinetic motion is large, into a cold sink, where the kinetic motion is lower. This is easy to visualize, the fast moving particles move into the areas of slow moving particles, until the two sources have an equal amount of movement. This dispels the common student misconception that objects “gain cold,” rather, the object loses heat by transferring it to the colder object, and in the process the average particle movement slows down resulting in a lower temperature.
Enthalpy is energy flow as heat transfer, specifically at constant pressure (Zumdahl & Zuhmdahl, 2010). Enthalpy is perhaps easiest to understand in terms of its units: Joules, the same as the unit for energy. Although a somewhat elusive definition, enthalpy is useful for quantifying changes in heat transfer during chemical reactions. In fact, the “heat of reaction,” or change in enthalpy, is defined as the change in enthalpy between products and reactants:
∆H = H
products
- H
reactants
A negative change in enthalpy means heat is released during the reaction, and a positive change in enthalpy means that energy is absorbed during the reaction. When heat is released, the change is considered to be exothermic. In exothermic changes, the system cools, while the surroundings heat up. Alternatively, when heat is absorbed during a reaction, the change is considered to be endothermic. In endothermic changes, the temperature of the system increases, while the surroundings lose heat, and therefore cool down.
Different substances require different amounts of heat transfer to raise their temperatures. For example, tile flooring changes its temperature much quicker than carpet. The carpet requires a lot of heat transfer to raise its temperature by one degree, whereas, the tile raises its temperature by one degree with much less energy input. This property is known as heat capacity (C), which specifically is the amount of heat absorbed per change in temperature:
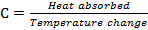
Heat capacity is different depending on the phase of a substance. For example, ice has a lower heat capacity than liquid water, meaning that a temperature increase of 1° in ice can occur with less energy than liquid water. In fact, the difference is roughly a factor of two, meaning ice can heat up twice as quick as liquid water can. Heat capacity is also usually defined in terms of amount of substance, called the specific heat capacity when measured for 1 gram of a substance, and molar heat capacity when measured for 1 mol of a substance (Zumdahl & Zuhmdahl, 2010). For example, the specific heat of water under ambient conditions is 1 calorie/(g
o
C).
Enthalpy can be very difficult to measure directly, but luckily using specific heat and temperature, we can calculate heat transfer much more efficiently. From chemical or physical change, we can measure the change in temperature of a substance, using a thermometer, as well as the mass of the substance, using a balance. From there, we can manipulate the definition of specific heat capacity to calculate the heat released or absorbed:
Heat = specific heat capacity × mass × change in temperature
If the measurements are made at constant pressure, then the measure of heat is equivalent to the measure of enthalpy (Zumdahl & Zuhmdahl, 2010).
3.
How Do Reactions Proceed Spontaneously? What Does Change Have to Do With Disorder (Entropy)?
(1 Lesson)
Spontaneous reactions are typically thought of as explosions, however spontaneous processes are defined as any change that occurs without outside interference—fast or slow. For example, the rusting of a car can occur over many years, but happens without interference, and is thus spontaneous despite how long it takes. Changes happen spontaneously when there is an overall increase in entropy (Zumdahl & Zuhmdahl, 2010). Entropy is a measure of disorder, the more organized something is, the less entropy it has. The second law of thermodynamics states that the entropy of the universe is always increasing. This means, that it is more likely to find a process in a state of disarray, than highly organized. For example, left unattended, a front lawn grows wild, it is much more unlikely to find grass evenly cut all the time. Spontaneity is moving towards disorganization, meaning that left unattended, the grass will grow wild because that is the state with more entropy, and therefore more disorder.
Entropy is dependent on a factor known as positional probability, among other things, which describes the probability of a specific arrangement of particles in happening. For example, take an arrangement of particles in a star shape, this could only have one specific arrangement, therefore the positional probability of the particles arranging in that shape are very low. On the other hand, there are many more ways to have particles arranged evenly spread throughout a room, therefore this arrangement has a much higher positional probability. The more spread out, the more disorganized particle are, the higher the positional probability for those arrangements are, and thus the more entropy they have.
Looking at the three phases, solids are typically more arranged than liquids or gases. Therefore, solids have the least entropy, while gases have the most entropy. Therefore, in a spontaneous change at higher temperatures, solids are likely to become liquids, and liquids gases.
Heat and temperature tie in closely to spontaneity. When heat is transferred into a system, the temperature usually rises. Temperature is a measurement of particle movement, and therefore when heat is transferred into a system, particles begin to move more rapidly. With a higher degree of particle movement, they are more likely to change their positions and formations. Thus, when heat is transferred into a system, the entropy in the system increases. The magnitude of the change in entropy also depends on the temperature. The lower the starting temperature, the more of an impact a given amount heat will have on increasing the random movement of the particles. Therefore, entropy (S) is directly related to the enthalpy (H; heat transfer at constant pressure) and inversely related to the temperature (T):
∆S = -∆H/T
Students can calculate the change in entropy by measuring the temperature, and by calculating the enthalpy change using specific heat (Zumdahl & Zuhmdahl, 2010).
4.
How Can Matter Change (Physical Vs. Chemical Change)?
(1 Lesson)
Matter can change in two ways: physically and chemically, depending on the identity of initial reactants and final products. In a physical change, reactants change forms but do not change identities on a molecular level. In addition, physical changes are usually reversible. The classic example is ice melting into liquid water. Both the solid and the liquid have the molecular formula H
2
O, the liquid can be refrozen into ice, and the ice can be melted back into liquid continually. On the other hand, chemical changes involve a rearrangement of atoms on a molecular level creating products with a different chemical composition. In addition, chemical changes are typically thought of as nonreversible.
In physical changes, particles can change how far apart they are, and how fast they are moving, without changing identities. For example, solids move much more slowly than liquids and gases. Furthermore, in physical changes, intermolecular forces, the weak forces between molecules, but not forces within molecules, loosen or tighten to hold particles farther or closer together, respectively.
Determining a chemical change requires the presence of a new chemical structure. The stereotypical signs of chemical change are taught as color changes, odor changes, gas bubble formation, precipitate (solid particles) formation, and heat change. However, some of these properties are not specific to a chemical change, for example liquid water can increase its temperature without changing its chemical composition. Similarly, a gas bubble formation is a physical change, as is solids falling out of solution to form a precipitate. These tests only sometimes show the formation of chemically new products, such as a new compound is formed, releasing hydrogen gas, or creating a new compound that is no longer soluble in the solvent. Thus, tests that show changes in chemical properties may be a better indicator of a chemical change. This can be done without advanced machinery such as measuring a change in color or taste (note: do not taste chemicals). A more advanced test could be measure the change in the bonds ability to absorb light waves, such as using spectroscopy.
Physical Changes
5.
How does matter change phases with regard to energy, enthalpy, and entropy during phase changes? (2 lessons)
A physical change does not change the chemical identity of a substance, just the location and the kinetic energy of the particles. A phase change is a physical change, as a single substance changes from its form, such as from solid to liquid or liquid to gas, or the reverse.
Each phase has its own associated enthalpy and entropy. Solids are typically compact and have strong intermolecular forces to keep them structured and highly organized, and thus low entropy. Because they are constrained in their position, the particles cannot move quickly, and thus have low levels of kinetic energy and temperature. As the phase changes from solid to liquid, then liquid to gas, the particles loosen their intermolecular forces allowing them to move more freely and thus with more kinetic energy. As kinetic energy increases, so does the temperature. The increasing disorganization in the change from solid to liquid, and liquid to gas, is paired with an increase in entropy.
It should be noted that there are other phases in addition to solids, liquids, and gases. Plasma is a phase of extremely high kinetic energy, and almost no intermolecular forces. Given the high amounts of kinetic energy, the temperature is also very high (Rayder, 2016). On the opposite side of the spectrum is bose-einstein condenstate—a state of matter where particles have no kinetic energy and have a temperature at -273K, absolute zero (Raizen, 2011).
When phases go from a highly ordered state (solids) to a less ordered state (gas), the entropy is increasing, and therefore making the phase change more favorable, and even spontaneous in certain circumstances. When phase change occurs in the reverse, condensation or freezing, enthalpy decreases as particles speed up and intermolecular forces are reformed
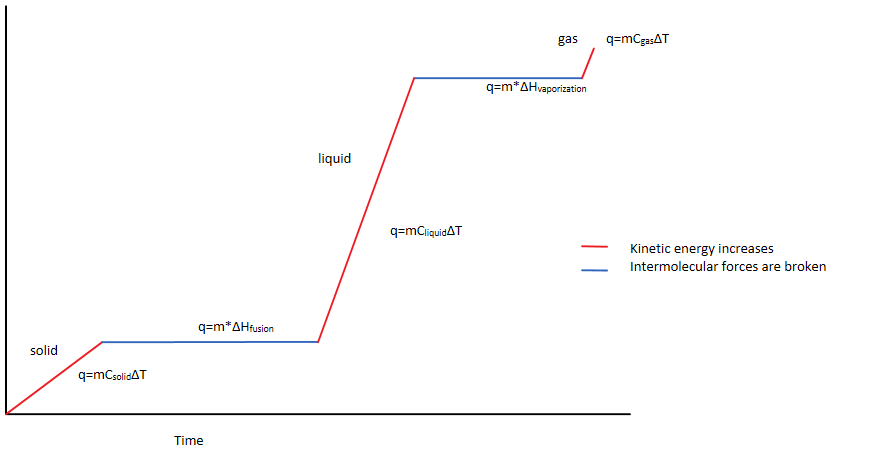
Figure 1 As the temperature increases over time, a solid changes into a liquid, then a gas. In different sections, the heat is being used to alternatively increase the kinetic energy or break intermolecular forces. The different quantifications are used in each section of the phase change.
Phase changes are a great way for students to practice calculation regarding enthalpy and specific heat capacity. For example, starting with a solid, the solid must first heat up before it can melt into a liquid. During this phase, the heat absorbed is used to increase the kinetic energy of the particles, as represented by the temperature increase (as seen in Figure 1). The heat is quantified with the equation for specific heat:
q=mC
solid
∆T
Where the heat (q) is quantified in the mass (m), specific heat capacity of the solid (C), and the change in temperature (∆T) (Nave, 2016).
Once the solid reaches its max temperature without changing phases, the phase change process begins. This time the heat is not being used to change the kinetic energy, and the temperature stabilizes (Figure 1). Instead, the heat is being used to break intermolecular forces. The heat can be quantified as:
q=∆H
fusion
x m
This time, heat (q) is quantified by the enthalpy of fusion (∆H
fusion
) and the mass (m). The process then continues from liquid to gas, this time using the specific heat of the liquid (C
liquid
) and the enthalpy of vaporization (∆H
vaporization
) (Nave, 2016).
Chemical Changes
6.
What are the conditions necessary to allow chemical change?
(1-2 Lessons)
Free energy is the energy that can be converted into work. For a reaction to occur spontaneously, that is for the entropy of the universe to increase, the free energy of a change must decrease (Shipman, 2009). We have already seen that reactions are more likely to happen when the entropy increases, or when entropy is positive, and when heat is released, or enthalpy is negative. The change in Gibbs free energy (∆G) is represented by the following equation:
∆G = ∆H – T∆S
The Gibbs free energy is a measure of how much enthalpy (heat transfer at constant temperature) occurs in a reaction as well as changes in entropy. Gibbs free energy is then based on two primary factors: Enthalpy (H) and Entropy (S), supported by Temperature (T).
This results in four possible scenarios (Table 1), in which entropy could be increasing or decreasing, matched with an enthalpy that could be increasing or decreasing. Spontaneous reactions, which is when ∆G is negative, will always occur in the scenario that entropy is increasing and enthalpy is decreasing, or is exothermic. Reactions can never happen spontaneously with the opposite conditions: decreasing entropy, and increasing enthalpy, or endothermic reactions (Zumdahl & Zuhmdahl, 2010). When only one of the two conditions are unfavorable, the likelihood of the reaction happening depends on temperature. If enthalpy increases, but entropy increases as well, the reaction will happen spontaneously only at high temperatures. And if a reaction decreases in entropy, but also decreases in enthalpy, the reaction will only proceed spontaneously at low temperatures.
Table 1. Signs of Enthalpy, Entropy, Gibbs Free Energy Direct an Endergonic or Exergonic Reaction
|
Enthalpy (∆H)
|
Entropy (∆S)
|
Gibbs Free Energy (∆G)
|
Endergonic or Exergonic?
|
|
–
|
+
|
–
|
Exergonic always
|
|
+
|
–
|
+
|
Endergonic always
|
|
+
|
+
|
+ @ low temperatures
– @ high temperatures
|
Low temp=endergonic
High temp=exergonic
|
|
–
|
–
|
+ @ high temperatures
– @ low temperatures
|
High temp=endergonic
Low temp=exergonic
|
As stated above, reactions that occur spontaneously release free energy (have negative ∆G). These reactions are referred to as exergonic. When reactions have the unfavorable thermodynamics, and are thus non-spontaneous, it does not automatically mean that the reaction can never occur under any circumstances. Instead, the reaction can occur, but only with the input of energy or work. Reactions that absorb energy are called endergonic (Shipman, 2009).
7.
How is Release and Absorption of Energy Dependent on Total Bond Energy? What is Activation Energy?
(1 Lesson)
Elements are held together within a compound by proximity of their outer electrons, or valence electrons. The elements share electrons forming bonds, the stronger the electron is held by another element, the more energy is required to break the bond. Total bond energy is a type of potential energy, storing the energy in holding electrons between atoms. When energy is absorbed by a substance it can be used to break intramolecular bonds. Conversely, when new bonds are formed energy is released. When there is more energy absorbed by a reaction to break bonds, than there is energy released in forming the new ones, the reaction is endothermic. When more energy is released in the formation of bonds than is absorbed to break the bonds, the reaction is exothermic. Nonetheless, all reactions need an input of energy to break bonds and begin the process (Shipman, 2009).
The energy used to start a reaction is called the activation energy. For a reaction to occur, the reactants need to align is a specific way, and collide with enough kinetic energy to set off a spontaneous change in positional arrangement into products. The prerequisites for a reaction to occur represent a barrier, and is known as the activation energy. At the infinitesimal moment at the maximum of the energy barrier is where the transition state between reactants and products is found. The transition state is incredibly unstable, and only in 2016 was the energy of a transition state first measured (Schmidt, 2016). Once over the energy barrier, a reaction can proceed, absorbing energy to break bonds, or releasing energy to form new bonds.
The progress of a reaction is graphed taking into account the initial energy of the reactants, the final energy of the products, and the activation energy to surmount the energy barrier (figure 2):
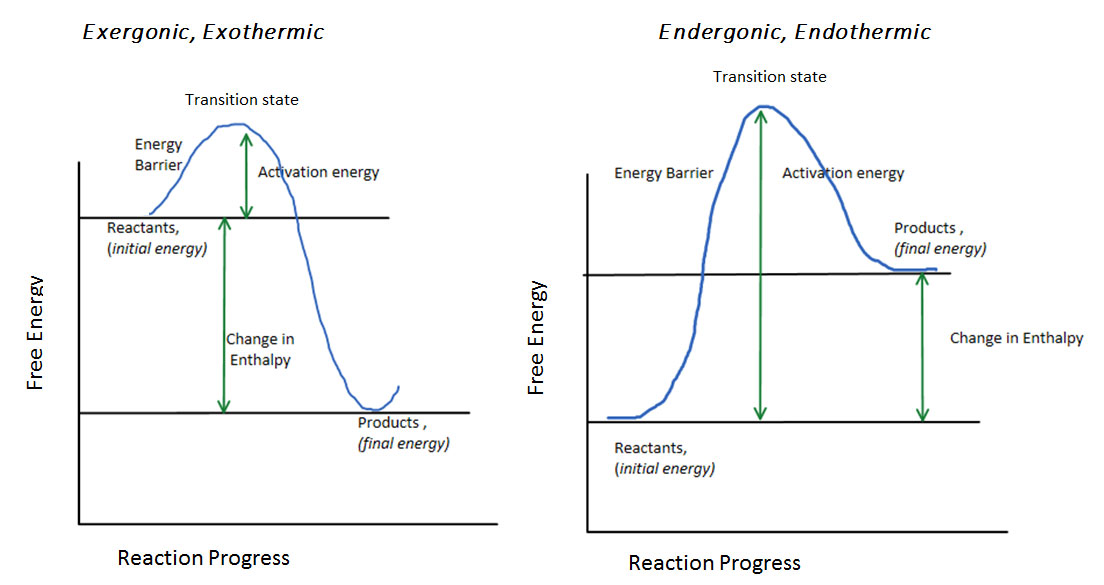
Figure 2 The reaction progress for an exothermic/exergonic reaction and an endothermic/endergonic reaction
The overall change in energy is an expression of the change in enthalpy (assuming changes in entropy are small). If the products have less energy than the reactants, as seen in the figure, then the reaction is exothermic because of the release of heat. When there is more energy in the products than the reactants, but both less than the activation energy, then the change in enthalpy is positive, and the reaction is therefore endothermic. The reaction progress can also be plotted with the y-axis depicting free energy, to show exergonic and endergonic reactions instead of exothermic and endothermic, respectively. Then any changes in entropy are also accounted for.
8. What affects the rate of reaction? (1-2 Lessons)
Although reactions may occur spontaneously, without outside intervention, the reaction may take years or centuries or longer, resulting a very slow rate of reaction. Gaining the activation energy to overcome the energy barrier is the defining factor in the reaction rate. Reactions may take years to reach the necessary activation energy to transition from reactants to products. For example, a mixture of H
2
(g) and O
2
(g) is stable indefinitely under ambient conditions, but if there is a spark, the reaction proceeds instantaneously. Therefore, factors that help reactants overcome the energy barrier between reactants and products increase the rate of reaction. These factors include: temperature, concentration, surface area, pressure, and the presence of a catalyst (Shipman, 2009).
Reactants must collide with enough kinetic energy to overcome the energy barrier. Kinetic energy of particles is measure in temperature, therefore, an increase in the temperature reflects an increase in the kinetic energy of the reactants (Shipman, 2009). With an increased temperature, reactants are more likely to overcome the activation energy barrier.
Reactants must collide with specific orientation to be able to make the transition from reactants to products. This specific ordering typically has low probability of occurring, which explains why the transition state has very low entropy. Increasing the concentration of reactants makes it more likely for reactants to bump into each other with the correct orientation because there are more reactants present with random orientations (Shipman, 2009). Similarly, increasing the surface area for a reaction involving a solid increases the positional probability, or the fraction of reactants that have the correct orientation for reaction to take place.
Manipulations of the pressure and volume of a system increases the rate of reaction for gas phase reactions. Increasing the pressure can increase the kinetic energy of the reactants, as seen in the gas law theory which shows that pressure is directly related to temperature for a given amount of gas in a given volume (Zumdahl & Zuhmdahl, 2010). By increasing the pressure, reactants have an increased force of collisions and at a greater rate. Meaning that more reactants likely have the energy to overcome the energy barrier. Similarly, by decreasing the volume, the reactants are more likely to hit each other due to space limitations.
The final reaction rate factor is whether or not a catalyst is present. A catalyst is a substance that increases the rate of reaction, but is not itself used up during the reaction (Shipman, 2009), and does not change the thermodynamics of the reactants and products. Catalysts, unlike other reaction rate factors, decreases the activation energy by holding reactants together in preferred orientations for the transition state.