What is Carbon and Why is Carbon Important?
If you were to explore the universe and count the different elements, hydrogen and helium would be the most abundant at about 74% hydrogen and 24% helium. These elements were produced during the “big bang” formation of the universe and make up about 98% of the universe as we currently understand it.6 Hydrogen is the lightest element with one proton and one electron. Helium is the second lightest element with two protons and two electrons. Heavier elements then these, such as oxygen, carbon, neon, and iron, were not formed during the “big bang.” Instead, they are created in stars through stellar processes and are much rarer, coming in at an estimated 2% of all the other elements combined! Carbon is in a very distant fourth place when it comes to the most common universal element, just behind oxygen.
Astronomers estimate that about 90% of the stars in the universe are in their main sequence stage, just like our beloved Sun, which by comparison is a medium-sized yellow dwarf star. A star exists in the main sequence stage for most of its life cycle, remaining in a constant state of balance between the star’s gravitational pull and the outward pushing force of energy released by nuclear fusion of hydrogen into helium at its core.7 Eventually, over billions of years depending on the mass of the star, that hydrogen source becomes depleted. When this happens, the nuclear reactions expand to consume hydrogen beyond the core as the star swells up to 400 times its original size, temperatures cool, and the star becomes redder in color. This is called the red giant phase.8 The same fate will befall our own Sun someday in an estimated five billion years. As the star’s atmosphere grows, its core contracts due to gravity. Temperatures and pressure increase as nuclear fusion resumes, this time using helium for fuel. The star condenses close to its original size. After billions of years of fusing hydrogen into helium, the helium will now be fused into other elements. When helium reaches a temperature of about one million Kelvin, three helium nuclei can fuse together to create a new element – carbon. If four helium nuclei fuse, oxygen is formed. Every element up to iron is created through various forms of nuclear fusion in stars. In fact, 115 of the 118 known elements are produced or sourced by stars in some way, either through continued nuclear fusion, cosmic rays, neutron star collisions, supernova, or radioactive decay.9
Upon the death of the star, most of the created elements are ejected into space. It is incredible to ponder that every past, present, and future living organism - from the tallest of trees, to the mightiest of whales, to the most miniscule of microbes – we all share a cosmic carbon connection with each other and the stars. In the vast expanse of space dominated by hydrogen and helium, carbon barely registers by comparison. But here on Earth, and by logical extension any other planet that is host to organic life, carbon is essential.
Every substance on Earth is made up of some combination of the 118 known elements found on the periodic table. Organic life, with few exceptions, utilizes about 25 of those 118 elements, but carbon, hydrogen, nitrogen, and oxygen are the most abundant and make up about 96% of living matter.10 Carbon is the element that binds them all together on a molecular level because of its ability to form stable bonds with many other elements, including itself. Carbon has an atomic arrangement of six electrons, two in a completed inner orbit along with four valance electrons available in its outer shell to form covalent bonds to other atoms or molecules. Each carbon atom can form 4 chemical bonds to other atoms, and the carbon atom is just the right, small size to fit in comfortably with larger molecules.
Carbon atoms can also share two or three electrons to form double or triple bonds. Carbon has an exceptional ability to bind with a wide variety of other elements and molecules due to its atomic electron arrangement. This property allows carbon to form a variety of large and complex molecules, including organic compounds like carbohydrates, lipids, proteins, and nucleic acids. Therefore, organisms on earth are all described as “carbon-based lifeforms.” Carbon is truly the foundation of all life on Earth and living things would not be able to live, grow, or reproduce without it. It is an element that is necessary to form complex molecules such as proteins and DNA. A human body is about 18% carbon, while the body of a plant is about 50% carbon.11 Carbon is a key ingredient in the food that sustains us, provides a major source of energy to power our planet, and the carbon in our atmosphere helps to regulate Earth’s temperature. Carbon is essential to life as we know it.
Elemental carbon can exist in one of three main physical forms, or allotropes, depending on its atomic structure. The known forms are diamond, graphite, and fullerenes. The ability of an element like carbon to crystalize in more than one configuration is known as polymorphism. Carbon also exists as an amorphous, or non-crystalline solid such as charcoal or soot. In a solid state, carbon dioxide is commonly known as dry ice. Under normal atmospheric pressure, solid carbon dioxide sublimates to carbon dioxide gas. Carbon dioxide can also reach a liquid state, but only under pressure above 5.1 atm. Pressurized liquid carbon dioxide is commercially used as a refrigerant.12
When carbon atoms are arranged in a three-dimensional tetrahedral array as a result of exposure to great pressure and heat, the result is a diamond – one of the hardest known minerals on Earth. Diamonds are formed naturally in the Earth over hundreds of millions of years due to the subduction of carbonate rocks such as limestone or dolomite to about 100 to 200 miles (or more) deep into the Earth’s mantle. This exposes the carbon in those rocks to approximately 725,000 pounds of pressure per square inch and temperatures between 900 to 1,300 degrees Celsius.13 There is a bit more to natural diamond formation that depends on how quickly the formed diamonds move through the mantle, but the diamonds that remain diamonds eventually make their way through the Earth’s crust and closer to the surface.
It is a misconception that diamonds are made from coal that has been subjected to heat and pressure. I recall Superman creating diamonds for Lois Lane in the 1978 film Superman by crushing several lumps of coal in his hands and using his heat vision. It would have been more accurate if he would have used graphite instead, since graphite is pure carbon while coal contains roughly 75% carbon and the rest are other substances or impurities such as hydrogen, oxygen, nitrogen, and sulfur. In addition, coal is formed from organic plant life that first appeared on Earth an estimated 500 million years ago.14 Natural diamonds with any embedded material (such as a garnet or another stone) can be dated by measuring the radioactive decay of those inclusions and researchers have found them to be between 1 to 3.5 billion years old, far outdating coal!
Carbon atoms arranged in a two-dimensional hexagonal lattice structure results in graphite. Graphite occurs naturally and is the most stable form of carbon under standard conditions of pressure and temperature. Although diamonds and graphite are the made of the same element, the atomic arrangement produces substances with vastly different hardness, color, conduction, and light refraction properties. Years before his execution in the French Revolution, prominent 18th-century French chemist Antoine-Laurent Lavoisier demonstrated that a diamond enclosed in oxygen and heated beyond 800 degrees Celsius will glow red, then white, and then a reaction between the carbon and oxygen will vaporize the diamond into carbon dioxide gas.15 There are some interesting YouTube videos demonstrating this phenomenon. For comparison, the temperature needed to burn the carbon of a diamond in regular atmospheric air is about 900ºC, while the flashpoint for wood and coal is about 300ºCelsius.
As a gas, carbon atoms are covalently double bonded to two oxygen atoms to form carbon dioxide (CO2). Carbon dioxide is an odorless, colorless gas at room temperature. It is also a greenhouse gas because of how it absorbs long wavelength energy and re-emits energy in all directions. About half of the absorbed energy is emitted out to space, but the other half is emitted back down to Earth. Although there are various processes that naturally remove carbon dioxide from the atmosphere, they take time. Researchers estimate about half to three quarters of atmospheric carbon dioxide is absorbed into ocean water within 20 to 200 years prior to human contribution of carbon dioxide.16 The rest can be absorbed through a variety of long-term geological processes that can take hundreds to thousands of years.
The Earth’s Carbon Cycle
Like Earth’s hydrologic (water) cycle, all the carbon on and within the Earth, its waters, its atmosphere, and its organic inhabitants is contained within a relatively closed system. The amount of carbon on Earth does not change significantly, but where on Earth that carbon is located does. Locations on Earth where more carbon is absorbed and stored than released are known as carbon sinks or reservoirs. Conversely, any process that releases more carbon than is absorbed is known as a carbon source. The movement of carbon through the carbon cycle is known as a carbon flux.
So how much carbon do we think is here on Earth? Scientists from the Deep Carbon Observatory (DCO), a network of over 1,200 multidisciplinary scientists from 55 countries, spent ten years from 2009 to 2019 assessing carbon reservoirs and fluxes to determine where carbon is stored and how carbon moves through the Earth’s carbon cycle. They currently estimate that Earth contains about 1.85 billion gigatons of carbon!17 The prefix “giga” means one billion. A digit of 1 followed by nine zeros, also written as 109. You may be familiar with some computer terms such as gigabyte or gigahertz, representing a billion computer bytes or a microprocessor that operates on a billion hertz frequency. To put the size of just one gigaton (can also be spelled gigatonne) of mass into perspective, a single gigaton is equivalent to one billion metric tons, or 2.2 trillion pounds, or approximately 10,000 fully loaded U.S. aircraft carriers! If you placed a one gigaton block of ice on top of Central Park in New York City so that it covered all 843 acres, it would extend about 1,120 feet high, almost as tall as the 1,250 feet height of the Empire State Building!18
The Deep Carbon Observatory now estimates more than 99% of the Earth’s carbon is below the surface, deep within the mantle and core, with most of it in the lower mantle. That’s 1.845 billion gigatons underground. Only 2/1000ths of 1%, a total of about 43,500 gigatons, is above ground.19 That includes the oceans, land, all organic life, and the atmosphere. About 85% of that, or 37,000 gigatons, is in deep ocean water – one of Earth’s major carbon sinks.20 Marine sediments, including the remains of exoskeletons and shell forming sea creatures, account for about 7%, or 3,000 gigatons, of the above ground carbon. Another 4.6%, or 2,000 gigatons is found in the terrestrial biosphere. The terrestrial biosphere includes the land surfaces and soil of the Earth, as well as every organism living on land, dead or alive, both plants and animals. The ocean surface contains about 2% of the near surface carbon, equivalent to 900 gigatons, and is a major carbon flux point for carbon exchange from the air to the ocean. Finally, there is our atmosphere, which currently holds 1.4%, or 590 gigatons, of Earth’s total carbon content.21 Please note that the atmospheric carbon estimate is strictly for carbon, not carbon dioxide.
The carbon cycle describes how carbon atoms continue to travel from the atmosphere to the Earth, then back into the atmosphere. Carbon reservoirs, also known as carbon sinks, are places where carbon is trapped or stored. Carbonate rocks such as limestone and chalk, the Earth’s oceans, soil and sediments, the Earth’s atmosphere, and plant life are all major carbon sinks. Learning about where carbon is stored, and in what amounts, is crucial to understanding the Earth’s carbon cycle. Carbon can be released back into the atmosphere naturally through volcanic eruptions, fires, and when organisms respire or decompose. Carbon is also released due to human activity, primarily the burning of fossil fuels and because of land development – especially including the production of concrete.22
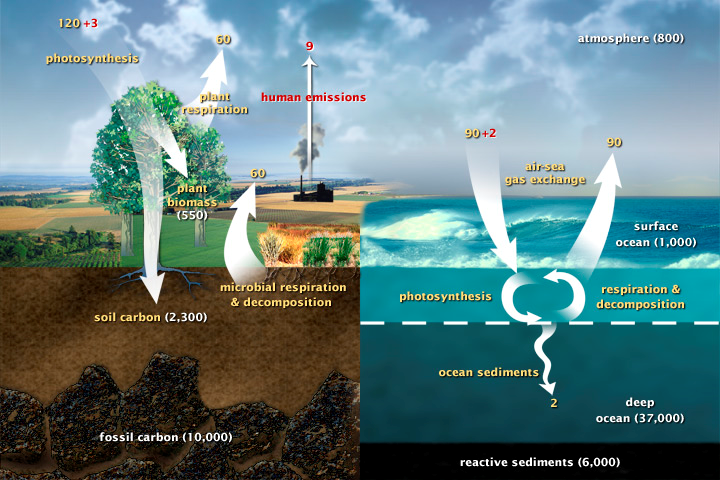
Figure 1: A diagram of Earth's fast carbon cycle that show the movement land, atmosphere, and oceans. Yellow numbers are natural fluxes, and red are human contributions in gigatons of carbon per year. White numbers indicate stored carbon. (Diagram adapted from U.S DOE, Biological and Environmental Research Information System.)
Earth has slow and fast carbon cycles. The slow carbon cycle can take 100-200 million years for carbon to move naturally between rocks, soil, ocean, and atmosphere. Rainwater containing weak carbonic acid promotes chemical weathering of rock, which releases carbon (in the form of bicarbonate), calcium, magnesium, potassium, or sodium ions that are carried to oceans. In the ocean, calcium and bicarbonate ions form calcium carbonate.23 This is the white residue that sometimes collects on faucets if you live in an area with hard water. In the ocean, calcium carbonate is made by shell-building organisms such as corals and plankton. Layers of shell and sediment sink to the seafloor and become cemented together over time, turning into carbon-storing sedimentary rock such as limestone. Eventually volcanic eruptions vent gases, including carbon dioxide, back into the atmosphere and new rocks are exposed by uplifting, bringing fresh silicate rock to begin the cycle again. There is also a faster component to the slow carbon cycle where surface water meets the air. Carbon dioxide gas dissolves in and out of surface water in a steady exchange with the atmosphere in balance with the carbon received through rock weathering. An increase in carbon concentrations in the atmosphere can result in the ocean absorbing more carbon dioxide. Once in the ocean, the reaction between water molecules and carbon dioxide releases more hydrogen, which makes the oceans more acidic.
While the slow carbon cycle can take hundreds of millions of years, Earth’s biosphere (all living things) cycles carbon at a much faster rate. Between 1,000 to 100,000 million metric tons of carbon moves through the fast carbon cycle per year.24 Plants and phytoplankton absorb carbon dioxide from the atmosphere and combine with water to form sugar and oxygen. Most plants, algae, and cyanobacteria utilize the process of photosynthesis to support metabolic activities such as growth, reproduction, and responses to the environment. The method of photosynthesis varies depending on the species, but the process always begins with the absorption of light through proteins containing green chlorophyll pigments. In plants, these proteins are found inside organelles called chloroplasts, while in bacteria they are in the plasma membrane. During photosynthesis, carbon dioxide enters the leaves of a plant through small pores called stomata. The roots of the plant absorb water and the plant uses the Sun’s energy (converted into chemical energy) to fuel cellular respiration that will synthesize carbohydrates such as sugars and starches from the carbon dioxide and water molecules. Some carbon is released back into the atmosphere through plant respiration. When that vegetation dies and is buried, some of that carbon remains in the soil. If it burns, carbon is released into the air as carbon dioxide. The fossil fuels like coal and oil that we uncover for energy production began as vegetation that eventually became buried deep within the Earth’s crust millions of years ago.
Carbon dioxide is returned to the atmosphere when plants break down to sugar to gain the energy for to grow, animals eat the plants and break down the plant sugar to get energy, plants die and decay (eaten by bacteria), or the plants are consumed by fire. In each case, oxygen combines with sugar to release water and carbon dioxide back into the atmosphere. In 1959, scientist Charles Keeling began taking atmospheric carbon dioxide measurements on the extinct volcano Mauna Loa in Hawaii. His results showed that carbon dioxide levels increased and decreased throughout the year to reflect the growing season. A more alarming concern was the realization that there was more carbon dioxide in the air than in the previous century and was steadily increasing.25 In 2003, the measurement milestone of 400 parts per million (ppm) was reached for the first time in over 55 years of taking measurements. This is similar to carbon concentration levels during the Pliocene Era (5.3 million to 2.6 million years ago) when the sea levels were higher and the average global temperature about 3ºC warmer than today.26
Carbon in the Air:
Carbon dioxide gas consists of one carbon atom and two oxygen atoms. Gases are typically measured by concentration in parts per million (ppm). It occurs naturally as a trace gas. It is odorless, colorless, and non-flammable. Earth’s atmosphere is 78% nitrogen and 21% oxygen. The remaining 1% is everything else, including argon (0.93%), carbon dioxide (0.04%), neon, and trace amounts of neon, methane, krypton, and hydrogen. Water vapor in the atmosphere can vary depending on location and time because air temperature determines the humidity capacity of air. At 30° C (86°F) a volume of air can hold up to 4% water vapor, while at 0°C and below (-32°F) a volume of air can only hold about 0.02% water vapor.27

Figure 2: The Keeling curve shows the ongoing rise in the atmospheric CO2 concentrations and the similarity of measurements taken on Hawaii's Mauna Loa (black) and at the South Pole (red).
Carbon dioxide is a greenhouse gas due to carbon’s ability to absorb and re-emit long wave radiation, much like a rock that retains and re-emits heat at night after a day in the sun. Nitrogen and oxygen thankfully do not share this characteristic. Air also contains tiny particles or droplets known as aerosols that remain suspended in the air. An estimated 90% of aerosols are from natural sources, such as mineral dust, sea salt particles, and partially burned organic carbon that can be sent aloft by wind, volcanic eruptions, or wildfires. The estimated 10% of anthropogenic, or man-made, aerosols include sulfur dioxide, nitrates, and smoke particles of organic and black carbon that have been released due to fossil fuel combustion and intentional biomass burning.28
It is unlikely that any carbon dioxide escapes or enters the Earth’s upper atmosphere to or from space. Earth’s atmosphere is held in place by Earth’s gravity, so carbon dioxide molecules would need enough energy to reach escape velocity.29 Smaller planets, such as Mars and Mercury, have a much thinner atmosphere due to their weaker gravitational force. Their atmospheric composition is different than Earth’s atmosphere because of it. Not only is the Earth an ideal distance from the Sun to support life, but it is also large enough to hold a protective atmosphere and magnetosphere.
Technically it is possible to gain carbon when objects from space collide with the Earth. And I suppose anything we have launched into space is carbon leaving, but that’s insignificant. There is no known steady, natural flow of carbon back and forth from space. Hydrogen and helium are light enough to eventually diffuse into space under several conditions. Certain interactions between Earth’s magnetic field and oxygen molecules also cause a small oxygen leak. Don’t worry, the Earth has enough hydrogen and oxygen to last at least a billion more years.
A carbon dioxide molecule has an atomic mass unit (amu) greater than the atomic mass unit of nitrogen and oxygen, making carbon dioxide denser that the surrounding gases. Atomic mass units are a form of measurement for atoms and molecules, just like the mass of a person may be expressed in pounds or kilograms. Atomic weight is determined by taking an averages of the element’s isotope variations. Isotopes are forms of the same element that differ according to the number of neutrons they have in their nuclei, although they still have the same number of protons. For example (and I am rounding amu values to the nearest tenth for simplicity), a carbon atom is about 12 amu, an oxygen atom about 16 amu, and a nitrogen atom is about 14 amu. Carbon dioxide is a molecule composed of one carbon atom and two oxygen atoms, giving it a total atomic weight of 44 amu. Oxygen molecules contain two oxygen atoms, for a total of 32 amu. A molecule of nitrogen gas is made of two nitrogen atoms, for a total of 28 amu. At 44 amu, carbon dioxide has a greater atomic weight than either oxygen or nitrogen gas. While these three heavier molecules are bound to Earth by gravity, lighter gases, like hydrogen and helium, can escape from Earth's gravity and eventually drift into space.
Despite being heavier, carbon dioxide does not form a settled, stratified layer under oxygen and nitrogen. Atmospheric gases are well mixed because of the force of diffusion as gas expands to fill a volume. There is also a lot of energy in the air due to uneven temperatures. Theoretically, if you could cool the Earth and enclose it in a sealed container until the only force acting upon it is gravity, the gases would settle in stratified layers.
What is the Greenhouse Effect?
The process that occurs when gases in the Earth’s atmosphere trap heat, much like the Earth under a blanket, is known as the greenhouse effect. Students may relate more to a blanket analogy rather than a greenhouse, but either way – it is a process that keeps the Earth warm. Without greenhouse gases in our atmosphere, the Earth’s average temperature would drop to as low as 0º Fahrenheit (14º Celsius).30 Earth would be an icy wasteland, hospitable only to organisms that could tolerate such conditions. Key to this understanding is the natural balance that makes our planet unique in our solar system with its ability to support an abundance of life. Too much greenhouse gas can have a detrimental effect. The planet Venus provides us an example of a planet where greenhouse gases have greatly increased the atmospheric and surface temperature of the planet. Too much heat would be trapped, and to continue the blanket analogy, it would be like wearing a winter coat under a large pile of blankets on an already hot day.
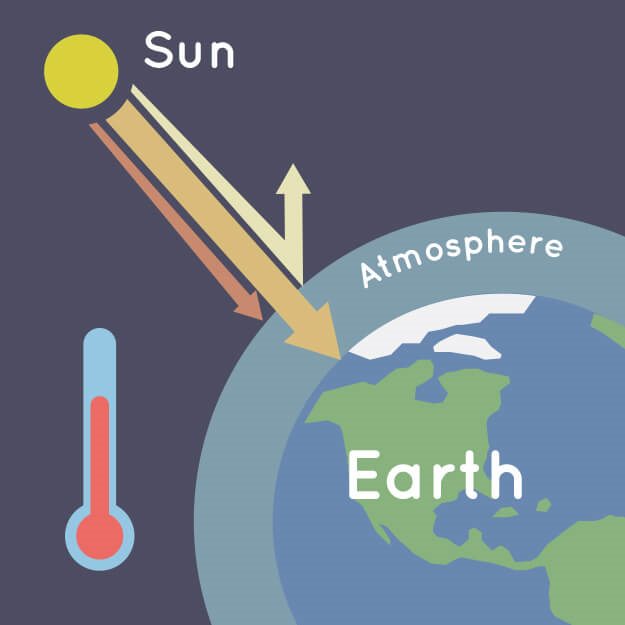
Figure 3: Earth's atmosphere traps some of the Sun's heat, preventing it from escaping back into space at night. Credit: NASA/JPL-Caltech

Figure 4: A greenhouse captures heat from the Sun during the day. Its glass walls trap the Sun's heat, which keeps plants inside the greenhouse warm - even on cold nights. Credit: NASA/JPL-Caltech
Energy from the sun is either reflected into space or absorbed by the Earth. Greenhouse gases are water vapor, carbon dioxide, methane, nitrous oxide, ozone, and chlorofluorocarbons (CFCs). They absorb the heat and radiate it out in all directions, very much like a stone that has been heated by the sun. Under the current balance, the amount of carbon dioxide has been increasing as human activity releases more and more of it into the atmosphere each year. This has led to an increase in temperature. Since warmer air can hold more water, this adds more water vapor to the atmosphere (and an increase in rainstorms in some regions).
A familiar concept that dark-colored objects left out in the sun get warmer than light-colored objects is a phenomenon known as albedo. Anyone who has walked barefoot on the white painted lines of a dark asphalt parking lot to reduce burning their feet is familiar with this concept. This has an impact on weather and climate on a planetary scale. In the Arctic regions, it can result in the melting (or build-up) of sea ice and glaciers. Sunlight is the primary driver of Earth’s climate with about 340 watts per square meter of energy from the Sun reaching the Earth. About one-third of that energy is reflected into space, and the remaining energy is absorbed by land, ocean, and atmosphere. Exactly how much sunlight is absorbed depends on the reflectivity of the atmosphere and the surface. Albedo can range between 0 (nothing reflected) and 1 (completely reflected like a mirror). Ocean water albedo is about 0.06, or 6 percent.31 Sea ice can range from 0.25 to 0.8, or 25 to 80 percent. Sea ice melts as global temperatures rise. This change to the Earth’s albedo as the result of melting ice further exacerbates climate change. The same can be said for man-made changes to the Earth’s surface such as removal of vegetation and adding building while paving of roads. Under current climate conditions, urban areas already experience a “heat island effect” where daytime temperatures are 1ºF to 7ºF warmer than outlying areas and nighttime temperatures about 2ºF to 5ºF higher.32
How Do We Know the Climate is Changing?
While weather describes the conditions at a particular time and place, climate refers to the weather conditions and patterns expected in a particular region as various times of year. Climate change refers to changes in the average conditions (such as temperature and precipitation) averaged over a long period of time in a particular region. Global climate change refers to the average long-term changes over the entire planet. Even before humans, the Earth’s climate has constantly been changing. For example, 20,000 years ago, much of North America was covered in glaciers but today we have a warmer climate and fewer glaciers. The Earth is used to these continuous changes over its 4.5-billion-year history and will continue to survive and adapt despite these changes.33 However, the disruption to climate as we know it could have some dire consequences. Some of the observable and measurable effects of climate change include rising sea levels, shrinking glaciers and Arctic ice, changes in temperatures that drive weather patterns, and changes in flower and plant blooming times. Earth’s climate is a large, interconnected system and the combined effects of these changes will impact one another.
We know what the Earth’s climate was like in the past by observing things that have been around a long time. For example, scientists can make observations about weather conditions over the years of a tree’s life by studying the insides of those trees. To study Earth’s climate even further back, hundreds of thousands of years ago, scientists (specifically paleoclimatologists) use things that have been around that long ago – icecaps, lake and ocean sediments. They study these by drilling out ice cores and sediment cores. Some ice cores can be several miles deep and each layer in an ice core (or a sediment core) tells scientists something about the Earth’s past.
We also know that added carbon dioxide, a greenhouse gas, to the atmosphere will create a growing feedback loop that results in warmer global temperatures unless humans intervene to reduce their collective global carbon output. Students having just studied energy resources are at a prime time to learn about the consequences of our energy demand and are ready to explore how science is used to determine energy resources that will meet our needs while doing the least amount of damage to the planet as we know it today.