Introduction
The first half of this unit will be an exploration of the major topics and themes of Nuclear Chemistry. In keeping with the sequential nature of chemistry, and nuclear chemistry being no different, our studies will follow a very orderly path.
Historical Perspective
The class will begin its studies on Nuclear Radiation, after having already examined the work of J.J. Thomson, Ernest Rutherford and Henri Becquerel, with a brief exploration of the life and work of Madame Marie Curie who, in 1911, was awarded the Nobel Prize in Chemistry for her work studying radioactive elements. Madame Curie is credited with furthering our understanding of radioactive materials. Sadly, her very life and death informed us of the risks associated with long term exposure to said materials. Marie Curie died in 1934 from Leukemia as a result of her work and exposure to radiation. We will continue exploring the history of Nuclear Physics by studying the work of Viennese born Lise Meitner and Otto Hahn.
Radiation
In this section of the unit, the class will study the mechanisms of discovery and the discoveries of Madame Curie; that radioactive materials "fogged" radioactive plates when exposed.
The unit plan is to introduce the concept of radioactive decay and the three main types of radioactive decay; alpha decay, beta decay and gamma radiation. In keeping with an already established tenet of science and this course, the Law of Conservation of Mass, we will write and track all particles associated with these types of emissions.

Nuclear Transmutations
We will then discuss transformations that occur as a result of nuclear decay. The attractive force, known as nuclear force, that is present to overcome proton-proton repulsive forces is sometimes insufficient to keep a nucleus intact. This event follows a fairly predictable pattern and can be appreciated quantitatively in a plot graph known as the
band of stability
, a concept that Lise Meitner intuited and paired to Einstein's mass-energy equivalence (E=mc
2
).
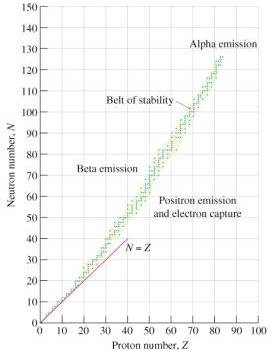
What happens to atoms when the nuclear force is overcome? In this section we will further explore the decay processes that result and put these processes into the context of a decay curve for various radioactive materials (a half-life curve).
Energy Yields in Fission and Fusion Reactions
We then explore the energy yields and circumstances under which fission and fusion reactions occur both naturally and as man-made events. Students compare and contrast fission and fusion reactions.

Fission Reaction
Fission reactions occur in such places as a nuclear reactor and a nuclear bomb. Students will compare and contrast the kinds of chain reactions that are controlled in a nuclear reactor and how chain reactions are uncontrolled in an atomic bomb explosion.
In addition to large energy yields, these processes yield nuclear waste which is a significantly hazardous material. Keeping these materials sequestered in the short and long term represents a significant engineering challenge to say nothing of the social and political implications of maintaining the integrity of that engineering feat.
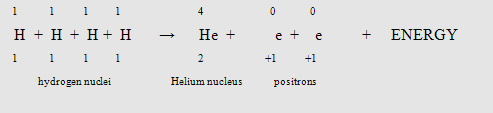
Fusion Reaction
Without nuclear fusion, life of as we know it could not be possible on Earth. The energy yields by our sun are experienced here on earth, some 96,000,000 miles away.
Practical Applications and Uses of Nuclear Materials
In this section we study the tools of the nuclear chemistry trade and applications of these materials in medicine. The circumstances surrounding Madame Curie's life and death provide ample and stark evidence as to the danger of being exposed to radioactive materials. These materials, however, are not without benefit. In this section students explore methods for detecting radiation exposure, applications of radioisotopes in medicine as diagnostic tools and as treatment for some diseases.