Roisin A. Macdonald
The intent of this unit is to have students explore the chemical and physical properties and changes that occur in the kitchen. Prior to proceeding with the intended subject there are a few things that need to be addressed and reviewed, the first of which is matter.
Matter
What is matter? By definition, matter is anything that has mass and takes up space. Everything around us, including ourselves, is made of matter. Matter can be classified into two primary categories: pure substances and mixtures. A pure substance cannot be separated by any physical means, pure substances are either elements like oxygen (O2), hydrogen (H2) and gold (Au) that contain only one type of atom, or compounds which contain two or more elements joined together in a definite proportion, like water (H2O), carbon dioxide (CO2) or sugar (C6H12O6). Elements cannot be broken down by physical or chemical means. Compounds can be decomposed or separated by chemical means, for example, in the process of electrolysis water molecules are broken down into hydrogen and oxygen atoms. Baking powder, which is baking soda (NaHCO3) plus a weak solid acid (cream of tartar), when added to warmed water releases carbon dioxide gas CO2 which creates the light consistency in many baked products.
Mixtures on the other hand can be separated by physical means. Mixtures contain two or more pure substances and can be further categorized by whether or not the substances are uniform within the mixture itself. A mixture is said to be homogeneous if the composition is uniform (components are not easily seen). Air, sugar water, stainless steel are all examples of homogeneous mixtures. A mixture is said to be heterogeneous if the substance composition is not uniform, that is you can see the components of the mixture. Plywood, salad dressing, granite, cinnamon and sugar are all examples of heterogeneous mixtures.
Another common way of categorizing matter is by its physical state, (commonly referred to as the phase of matter) that is, a solid, a liquid, gas, plasma or the most recent state of Bose-Einstein condensates. For the sake of simplicity, we will focus on the first three, most common states: solid, liquid and gas.
All matter is composed of molecules, it is how these molecules are arranged and move in reference to one another that determines the substances physical state. In the gaseous state the molecules are free to move about, that is the gas molecules take on no definite shape or volume. The molecules within a gas have no specific arrangement and are well separated. Due to the space between the molecules of a gas it is easily compressed. A substance that is in a liquid state has molecules that are closer together, but still in a random pattern, the substance still has no definite shape. Like gases, liquids take on the shape of the container holding them. The molecules in a liquid can easily slide past one another but are much closer together compared to a gas. Finally, a solid has molecules arranged so that the solid has a definite shape and volume. The molecules are arranged in a more rigid pattern and vibrate only slightly; there is little room for the molecules to move from place to place as in the liquid and gaseous states. A solid is not easily compressed since its molecules are naturally close together and there is little room left for compression.
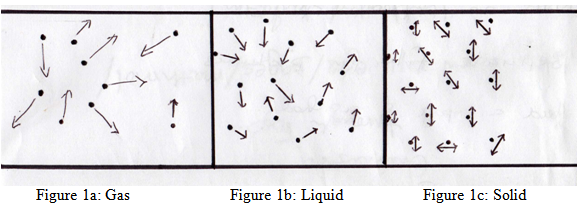
Let us take for example, water molecules. We all know water can easily exist in all three states dependent upon the temperature, a gas as steam, as liquid water, and as a solid, ice. Figure 1 illustrates the molecular structure of the water molecules in each state. Figure 1a, depicts the water molecules in a gaseous state, the molecules have plenty of space between them and are free to move about. Figure 1b shows the molecules of liquid water, note the molecules are more closely arranged, you can see how the molecules are able to slide past one another but are confined to a specific volume and take on the shape of the container. Lastly, Figure 1c illustrates how the molecules of solid ice are arranged in a definite pattern, the ice molecules can only vibrate in place not slide past one another, as in the liquid state or bounce off each other as in the gaseous state. Water in the solid state has a definite shape and volume.
Lesson one of this unit, Movin' Molecules, allows students to see firsthand how the movement of the molecules becomes more restrictive as they move from a simulated gaseous state down to a simulated liquid state and finally confined to a small area depicting the solid state.
Physical and Chemical Properties
The next item that needs to be discussed is the difference between physical and chemical properties of matter. First, let's establish the fact that a substance's state of matter is a physical property. A physical property must be easily observed and cannot culminate in a new substance, i.e. the composition of the substance remains the same. Physical properties are used to describe the substance. A substance's size, color, shape, and texture are all physical properties since they are used to describe that substance. Other examples of physical properties are the substance's melting point, boiling point, viscosity, solubility and density. The main characteristic to remember regarding physical properties is that the substance does not change; water is still water whether it is a solid, liquid or gas, its density is always one gram/milliliter (at room temperature), its melting point is always zero degrees Celsius and its boiling point is always one hundred degrees Celsius (at standard atmospheric pressure).
One physical property middle school aged students have trouble grasping is density. Density is defined as mass of an object per unit volume. The density of water is one gram per milliliter (at room temperature). Densities are often shown as a comparison to water, that is either the substance is more dense (density >1 gram/milliliter) and therefore sinks in water or less dense compared to water (density 1 gram per milliliter) which will naturally float. This concept can be demonstrated with a piece of balsa wood (less dense) and a rock (more dense) in water. The table below lists the density (1,2) of some common substances:
Balsa wood 0.2 g/cm³
Milk 1.03 g/cm³
Lead 11.0 g/cm³
Iron 7.8 g/cm³
Ice (0°Celcius) 0.92 g/cm³
Water (20° Celsius) 1.00 g/cm³
Gold 17.2 g/cm³
Silver 9.3 g/cm³
Use of the example above in the classroom may lead to the question, "if water and ice are the same substance, why does ice float in water?" The answer is quite simple, the structure of the ice molecules are arranged as a crystal lattice, the ice molecules within the lattice take up more space compared to the molecules in liquid water. In other words, as water freezes it expands, the crystal lattice has more volume (takes up more space), so that a liter of ice weighs less than a liter of water. As noted above, the density of ice is slightly less than the density of water; the heavier water displaces the lighter ice so it floats. This is the reason why water in rivers and lakes freeze at the top but is still liquid underneath, the less dense ice floats on the top.
Determination of Density using a Twinkie
Density is calculated by measuring the mass of an object and then determining the volume either through direct measurement (by liquid displacement) or geometrically, through mathematical measurements and calculations. Determining density is then a simple calculation of mass divided by volume. In keeping with our theme of food, lesson two is how to determine the density of a Twinkie, as a side bar the students can also calculate how much of the Twinkie is actually air (3).
The mass of the Twinkie is determined using a triple beam balance or a laboratory digital scale. The unit customarily used for measuring mass is grams. The shape of a Twinkie is nearly rectangular prism; therefore we will utilize the mathematical formula for the volume of a rectangular prism to determine the volume of the Twinkie:
Volume = length x height x width = cm
3
The second method used to determine volume is the water displacement method. This method can be traced back to around 250 BC when the Greek mathematician, Archimedes was assigned the task of determining if King Heiro I of Syracuse was being swindled out of his gold by his goldsmith. As the story goes, The King Heiro I of Syracuse noted his goldsmith seemed to be living a lifestyle much higher than he was being paid. King Hiero suspected the craftsman was stealing his gold by substituting silver when crafting his royal crowns. Archimedes knew that gold was heavier than silver so all he had to do was to determine the density of the two crowns. Massing the crowns was no issue but how was he going to get the volume. Archimedes could melt down the two crowns to make cubes and calculate the volume that way, but that would mean ruining the crowns, not such a good idea. One day, as Archimedes was getting into the bath he noted the water spilling out as he entered. Archimedes theorized that the amount of water displaced out of the tub was directly related to the volume his body took up in the tub. He jumped out of the tub running naked in the streets yelling "Eureka! Eureka!" Greek for "I have found it!. By measuring the water displaced by each of the crowns he determine the volume of each; calculating the density he proved the goldsmith was using silver and gold not just pure gold because the gold alone was more dense than the silver gold combination(4).
As the lesson shows students how to calculate volume both geometrically and by water displacement method, you can discuss the possible causes of error between the two methods. In the geometric method we are actually squaring off the rounded edges of the Twinkie, therefore, we would expect the volume as calculated from geometric measurements to be slightly more than the volume obtained through the direct measurement water displacement method. At the end of the lesson the students can pool their results to calculate the class average for mass, volume and density, this will give them the opportunity to utilize mathematical averages which you may or may not have to guide them through.

Getting back to the Twinkie, volume can be determined the same way as Archimedes did it, submerge the Twinkie in a known amount of water and determine the volume of water displaced. When doing the water displacement test for volume, be sure to leave the Twinkie wrapped, remove the packaged air by making a small hole in the wrapping, gently pushing out excess air and resealing the package prior to submerging, this will give us the actual volume of the Twinkie. As you know the Twinkie is light and fluffy which is part of its appealing taste. So how much of the Twinkie is cake/cream and how much is air? Since we already know the volume of the Twinkie as made we will just need to measure the volume with the air removed. To do this we will liquefy the Twinkie in a blender and measure the volume of the Twinkie puree using a graduated cylinder (see photo to the left). It is then a simple calculation of:
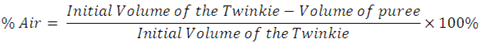
For reference, when I did the experiment I found the volume of the Twinkie through water displacement to be 140 milliliters compared to 143 milliliters utilizing the geometric mathematical method. The volume of the puree was found to be 42.8 ml, making the percentage of air in the Twinkie 69% (based on the 140 ml volume as determined by water displacement method). These values of course will vary slightly from group to group but the averages should be relatively close. For reference, I found the mass of the Twinkie was 44.5 grams with the density of the Twinkie calculated as 0.32 g/cm³. The object of the lesson is to have students learn the basic principles behind the measuring of density and determination of volume using two different methods.
Chemical Properties & Reactions
Chemical properties of matter are observed through chemical reactions; that is, the original substances, or reactants, combine or are separated chemically to form a new substance or substances called products. Take for example, the making of Monkey Bread at the end of lesson three, all of the ingredients are mixed together (exhibiting physical reactions/changes) and then the bread is baked which culminates in a new product which is, by definition, a chemical reaction. The overall concept to be remembered when deciding whether or not a change is a physical or chemical one is to determine if a new product has been made. Chemical change is a term that is synonymous with chemical reaction. In order to have a chemical change in a substance there must be a chemical reaction. This unit deals with chemical and physical properties and changes as they relate to baking treats.
Leavening Agents
What are leavening agents? Leavening agents are the components of a recipe that render the final product light and fluffy. There are three basic kinds of leavening agents, mechanical, chemical and biological. Leavening agents react with the moisture, heat and other ingredients in the recipe to produce a gas that becomes trapped in the batter or dough. It is this gas, upon heating, that gives rise to baked products.
Mechanical leavening involves the use of air or steam. Air is introduced to the batter or dough through beating, whisking or mixing, the air is then released during baking. Steam on the other hand is generated by the water in the batter that is converted to steam upon heating during the baking process. The trapped steam expands giving rise to the baked product.
Popovers and cream puffs are both good examples of mechanical leavening. Basically making popovers is a very simple recipe of flour, eggs, milk and a dash of salt, the air is incorporated mechanically while beating the ingredients together, the trapped air produced while mixing and the steam generated gives rise to the popovers as they are baked in a hot oven. Another example of mechanical leavening is cream puffs. Cream puffs combine water, butter, flour and eggs, again the steam produced by the water and air incorporated during the mixing is released during the baking process.
The second type of leavening agent is chemical. Chemical agents give rise to the baked product when the chemical agent reacts to produce a gas (usually CO2) which becomes trapped and then released during the baking process. The two chemical agents we will focus on are the most common ones, baking soda and baking powder. Any student who has ever made chocolate chip cookies or helped bake a cake has some previous knowledge of these "chemicals". Baking soda, sodium bicarbonate, is a mainstay in most homes, it is used as an abrasive cleaner, put in the fridge to absorb unpleasant food odors, flushed down the sink to clean and deodorize the pipes it can even ends up in your toothpaste helping you keep your pearly whites-white. So what is exactly happening? The gas we are looking to produce is CO2, carbon dioxide. Baking soda is sodium bicarbonate that reacts with a weak acid solid like cream of tartar according to the following equation:
NaHCO3 + H ' Na + H2O + CO2
Sodium Hydrogen ion ' Sodium + water + carbon dioxide
Bicarbonate (from weak acid)
Most recipes today call for the use of baking powder which is just baking soda with the addition of a dry acid (cream of tartar) and starch. The acid in baking powder is inactive until it is mixed with water. The reaction of the sodium bicarbonate and the dry acid is relatively quick which is why a lot of recipes have you mix dry ingredients separate from the wet ingredients. Most baking powders are double acting leaveners, that is, the initial reaction noted above along with mechanical leavening gives rise to the batter at room temperature while the addition of sodium aluminum sulfate reacts only at an elevated temperature to cause the cake to rise while in the oven.
The third type of leavening agent is biological. Yeast is a unicellular fungi that can produce its own food releasing CO2 and alcohol as waste products. The production of carbon dioxide is the result of the combination of yeast with sugar water. The chemical reaction that allows yeast to feed on simple sugars to produce energy needed for growth is called fermentation. The fermentation reaction is as follows:
Yeast + C6H12O6 2CH3CH2OH + 2 CO2 + energy
Glucose Ethyl alcohol carbon dioxide energy needed to reproduce
As seen in the reaction above, carbon dioxide and alcohol are the waste products produced by the fermentation process. These wastes are what bakers have taken advantage of for thousands of years to make light, fluffy leavened bread. The yeast and alcohol become trapped in the dough as bubbles. The dough batter is then mixed with flour where the starch in the flour forms a matrix supported by proteins. Upon baking the trapped gases evaporate leaving holes in the bread.
The last lesson in this unit allows students to see firsthand, the production of CO2 utilizing active dry yeast and sugar water. Prior to doing the experiment you may want to do a demonstration using active dry yeast added to warm sugar water (1 tablespoon of table sugar to 1 cup of water heated to approximately 105°F) in a glass Pyrex measuring cup, as shown or in a glass beaker. The first photo below shows the initial reaction where the yeast is beginning to bud, approximately 2-3 minutes after mixture.

Yeast & Sugar Water Reaction at 2-3 minutes
The photo below is the same yeast/sugar water after the yeast has bloomed (reproduction) by fermentation (after fifteen minutes).

Yeast and sugar water reaction after 15 minutes
For reference, yeast production is temperature dependent, that is, at temperatures above the yeast will die, too cold and the reaction will not proceed. To emphasize this concept incorporate ice water and boiling water to the demonstration asking the students to predict what they think the outcome for each will be and have them explain why.