Roisin A. Macdonald
Objective
-
1 .Students will be able to define the term density.
-
2. Students will determine volume of a Twinkie both geometrically and directly through
-
water displacement method
-
3. Students will measure the mass and volume of a Twinkie to calculate density
-
4. Students will determine the percentage of air in a Twinkie (Teacher lead demo)
Materials:
Demo: 600 milliliter beaker; 400 milliliters water; black permanent marker;
Piece of Balsa wood; (2) Rocks
Student activities: Twinkies; balance; 1000 milliliter beaker; 50 ml graduated
cylinder; blender (teacher operated); Metric ruler; permanent marker; calculator
Procedure:
Demo: The teacher will introduce the lesson by asking students if anyone knows what we mean by density. After some discussion define density as mass (grams) per unit volume (milliliter or cubic centimeters). Explain that often chemists refer to density of an object compared to water. Show students the block of wood and the rock and ask which is more or less dense than water. Demonstrate that balsa wood is less dense than water because it floats and the rock is more dense because it sinks. Ask students how they might determine the density of a rock given a balance, beaker with water and a graduated cylinder; facilitate a discussion that leads to massing the rock with the balance and measuring the displaced volume of water, with the data collected, calculate the density.
Student activity I:
|
1.
|
Have students get into groups of two or three.
|
|
2.
|
Hand out the instruction and data sheets to each group (see end of lesson)
|
|
3.
|
Review the basic instructions on calculating volume of the Twinkie by measuring the length, width and height of their Twinkie.
|
|
4.
|
Give each group (1) Twinkie (have extras so everyone gets to have one after the lesson)
|
|
5.
|
Have students calculate the volume of their Twinkie (length x width x height)
|
|
6.
|
Have students measure the mass of the Twinkie using the balance
|
|
7.
|
Students can then calculate density by dividing mass by volume.
|
|
8.
|
Students can then eat their Twinkies
|
Student activity II: Determination of volume of a Twinkie using the direct, water displacement method. (see student worksheet at the end of the lesson)
Teacher lead activity to determine percentage of air in a typical Twinkie based on the volumetric difference between the calculated volume of a standard Twinkie compared to volume of the Twinkie after the removal of air.
|
1.
|
Measure the length, width and height of a standard Twinkie; calculate the volume; record as V1
|
|
2.
|
Take a Twinkie and 5 milliliters of water place into a blender and puree (to remove air)
|
|
3.
|
Carefully transfer the Twinkie puree into a 50 ml graduated cylinder , record the volume of the Twinkie without air as V2a. Final volume V2 is equal to V2a minus 5 milliliters of water added to help liquefy the Twinkie.
|
|
4.
|
Have students determine the percentage of air in the Twinkie using the following formula:
|
V1-V2/V1 X 100% = % air in a typical Twinkie
Student Instruction/Data Sheet:
Today we will be measuring both the mass and volume of a Twinkie, we will then calculate the density.
|
I.
|
Determine the Mass of a Twinkie
|
-
Materials:
-
Twinkies
-
Triple beam balance
-
-
Procedure:
|
1.
|
Unwrap the Twinkie
|
|
2.
|
measure the mass of the Twinkie in grams using the triple beam balance
|
-
and record the mass on your data sheet
-
|
II.
|
Volume of a Twinkie: displaced water method
|
-
Materials:
-
1000ml beaker with 50 ml increments
-
500 mls water
-
Pin
-
tape
-
50 ml graduated cylinder
-
10 ml graduated cylinder
-
Permanent marker
Procedure:
|
1.
|
Put 500 mls of water into the 1000 ml beaker
|
|
2.
|
With a pin, poke a small hole in the top of the Twinkie wrapper and carefully push out the air.
|
|
3.
|
Reseal the Twinkie package covering the pin hole with tape
|
|
4.
|
Submerge the Twinkie into the water. While the Twinkie is submerged mark (with permanent marker) the new water level on the beaker
|
|
5.
|
Remove the Twinkie
|
|
6.
|
Using the 50 ml graduated cylinder add water back into the beaker up to the water level as marked (you may need to utilize the 10 ml graduated cylinder for the last few milliliters needed to get to the mark)
|
|
7.
|
Record the final volume on the data sheet as Volume (with Twinkie)
|
III. Volume of a Twinkie: Geometric calculation
Materials:
|
1.
|
A Twinkie
|
|
2.
|
Metric Ruler
|
-
-
Procedure:
|
1.
|
Remove the Twinkie from the wrapper
|
|
2.
|
Using a metric ruler measure the height, width and length of the Twinkie
|
|
3.
|
Record each value in centimeters on the data sheet
|
|
4.
|
Calculate the volume of the Twinkie using the following equation:
|
-
Volume = Length (cm) X Width (cm) X Height (cm)
IV. Density of a Twinkie
Density is defined as mass per unit volume. To find the density of the Twinkie
use the following formula:
Density = mass of the Twinkie (grams) / Volume of the Twinkie (cm³)
Student Data Sheet:

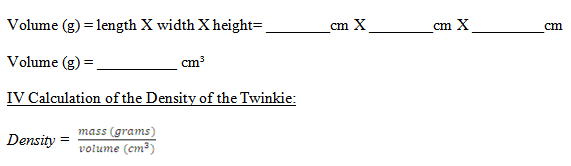
You may use either volume obtained in the experiment above, i.e. the volume calculated from the geometric measurements Volume (g) or the volume obtained directly from the water displacement method V(wd).