During the first quarter, phases and phase changes are covered in the phychem curriculum. Discussions are focused on phase changes among solids, liquids and gases also whether energy is increasing or decreasing during the phase change. Students are asked to learn not just phases and phase changes but molecular spacing and molecular motion in the three phases. In solid, molecules are close together and do not move relative to one another. In liquid, molecules are moderately close with limited motion, while in gas, molecules have the largest distance between each other and with the most molecular motion of the three phases. For the six phase changes there is either an increase or decrease in thermal energy between the starting and ending phase. Melting, evaporation and sublimation require an increase in molecular motions. Freezing, condensation and deposition require a decrease in molecular motions. The idea here is that all matter fundamentally carries some motion explained by the Kinetic Theory of Motion. This theory states that all matter (solid, liquid, or gas) is composed of small particles that are in constant motions
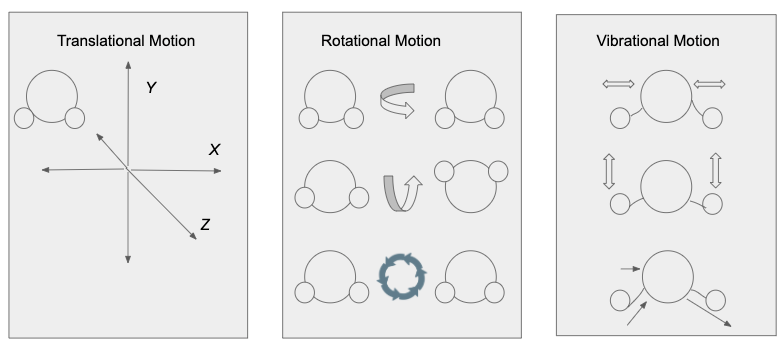
Figure 4: The translational, rotational and vibrational motion for a water molecule are depicted in this figure. 23
Translational, rotational, and vibrational motion are the three types of motions that molecules can exhibit. Translational motion is the movement of an atom or molecule along the x, y, or z coordinates. Rotational motion is when the whole molecule rotates on an axis. Moreover, vibrational motion is the repetitive motions that change the bond lengths or angles in a molecule.
Molecules are limited in the types of motion exhibited by the number of atoms in the molecule. One example is diatomic molecules that are molecules with only two atoms. These molecules can display translational, rotational, and vibrational motion. The translational motion may be experienced along the x, y, or z axis and this means that there are three degrees of freedom for translation in a diatomic molecule.24 Rotation has two degrees of freedom and vibration for a diatomic molecule is one. If all of these are possible ways that a diatomic molecule can move are added together we get a total of 6 degrees of freedom. Another way to calculate the degrees of freedom is by multiplying three with N, where N is the number of atoms in the molecule.25
We can apply all of our wave, kinetics, and phase change knowledge to a real-world experience making popcorn. Inside each kernel, there is a small amount of water. When the water in the kernel absorbs the microwave radiation, the frequency of the radiation is in resonance with the natural rotational frequency of water molecules. Thus, the rotational motion of the water molecules can be excited with higher kinetic energy. The increase in kinetic energy heats up the water at a temperature that turns liquid water into steam. Steam takes up more space than liquid water and thus the popcorn kernel pops.