The uptake of anthropogenic atmospheric carbon dioxide (caused or influenced due to human activity) is changing the ocean’s chemistry13. pH is a measure of how acidic or basic the water is. It is also a measure of the amount of free hydrogen and hydroxyl ions in water. The pH scale ranges from 1-14, where 7 is considered neutral, anything lower than 7 is acidic and more than 7 is basic or alkaline. Acidity is a measure of the concentration of free hydrogen ions (H+) in a water-based solution. So, the increase in free hydrogen ions (H+) causes high acidity or lower pH. One needs to understand that addition of CO2. decreases the pH of the ocean waters. On the other hand, the removal of CO2. increases the pH of ocean waters. This decrease in the pH makes the ocean water acidic. This phenomenon is called ocean acidification (OA). This process is slowing down the marine animals from making the skeletons and shells and is also negatively affecting the plants, animals, coral reefs, and the entire marine ecosystem.
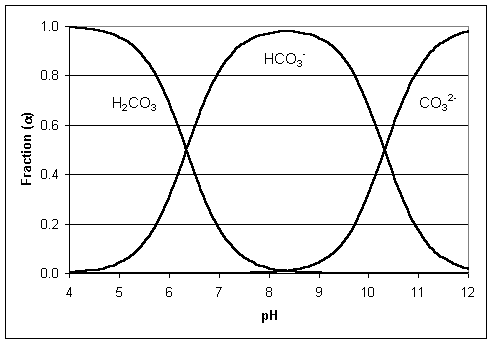
Figure 3: Ionization fraction plot of CO2
The Revelle Factor (buffer factor) is a measure of the “oceans buffer capacity for the carbonate system in sea water and fresh water”14. It is also a good indicator of the oceans pH levels. With the increase in the absorption of the anthropogenic CO2. Figure-3 explains the ionization factor (𝛼) of CO2 and how it affects the ocean pH. It is also a good indicator of the pH levels of ocean water. The process of ocean acidification (OA) is complicated and could be explained as follows (see Figure-4). It starts with the absorption of anthropogenic carbon dioxide (CO2) from the atmosphere by the ocean. This increases the dissolved inorganic carbon concentration (DIC) at the surface of the ocean waters. Thus the accumulation of atmospheric CO2 causes the following changes in the ocean water chemistry. First, the CO2 gas is converted into CO2 (aq)15. The CO2 (aq) can also dissolve limestone, which is a sedimentary rock made up of calcium carbonate (CaCO3)16. Many marine animals also use calcium carbonate (CaCO3) to make their shells, exoskeletons, and other structures. Next, the CO2 (aq) reacts with water to form carbonic acid (H2CO3) which is a weak acid. The carbonic acid (H2CO3) makes the ocean waters slightly acidic by increasing the number of free hydrogen ions (H+). Then the carbonic acid (H2CO3) thus formed can dissociate into bicarbonate (HCO3-) and free hydrogen ions (H+). Lastly, the bicarbonate (HCO3-) can further dissociate into carbonate ions (CO3-2) and more free hydrogen ions(H+). The above reactions are all reversible, meaning, they can also take place in the opposite direction (represented by arrows pointing in both directions) thus changing the chemistry of the surface waters of the ocean. Given that most marine organisms live in the upper waters of the ocean, these above changes in the chemistry of the water have devastating effects on the daily survival of these organisms.
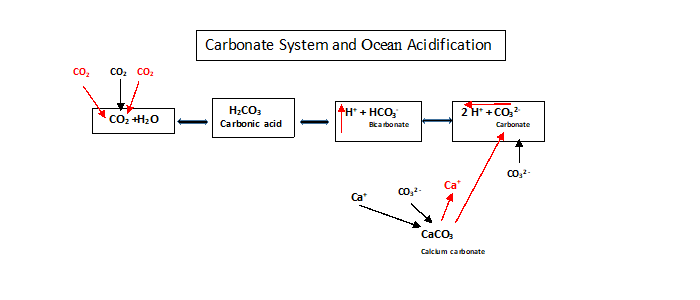
Figure-4: Carbonate System and Ocean Acidification (Adapted from graphics created by Chris Gobler, Stony Brook University)
According to the United States Environmental Protection Agency (EPA)17, during the pre-industrial times the average pH of the oceans was approximately 8.2 and today the average pH is around 8.1. Given that the pH scale is logarithmic. Thus decrease in one unit of pH represents approximately a 10 fold increase in the acidity of the ocean waters. CO2 forms a weak acid in water know as carbonic acid (H2CO3). That said, the increase in the absorption of anthropogenic CO2 (sink) from the atmosphere into the oceans is resulting in the decreasing pH of the oceans. The scientific community is alarmed that the atmospheric CO2 has increased 40% higher than the pre-industrial time and the current ocean acidity, on average, is roughly 25% higher than what it was during the pre-industrial times.
According to scientists, approximately 26 percent of all the anthropogenic CO2 released from human activities such as burning fossil fuels, changes made to the landmasses, cement production, so forth is being absorbed by the oceans. This process has been intensifying since the beginning of the Industrial Revolution in 1760. According to the Global Carbon Project18, fossil fuel burning account for approximately 35 billion tons of CO2 produced, and deforestation in the tropics adds roughly 5.5 billion tons which are some of the sources of CO2. Out of the CO2 thus produced, only half stays in the atmosphere causing global warming, while a quarter of it is taken up by plants and trees and the other quarter is absorbed by the oceans. Research studies stress that the oceans can hold up to 50 times more carbon than the atmosphere. As the concentration of CO2 in the atmosphere increases, so does its absorption by the oceans. This process is disrupting the chemical balance of ocean waters.
Metaphorically, some of the negative effects of ocean acidification19 are referred to as the Osteoporosis of the Sea20. According to medical professionals, osteoporosis (porous bone)21 is a bone disease where the human body loses too much or makes too little bone. This process weakens the bones and makes the body prone to accidents and injuries. This condition results in a fragile skeletal system and creates pores or honeycomb-like structures in the bones. It also decreases bone density, which makes the bones fragile and easy to break. In the same fashion, ocean acidification is negatively affecting the shell and skeleton formation of marine plants and animals. Marine animals need carbonate and calcium to form their shells. Thus the addition of the anthropogenic CO2 to the ocean waters is decreasing the carbonate concentrations. Therefore, the process of ocean acidification is decreasing the availability of carbonate, slowing down the process of shell formation, and dissolving the existing shells. The decreasing rate of shell formation and increasing rate of shell dissolution is therefore threatening the existence of marine ecosystems.
Current research stresses that the ocean surface temperature is rising. The main culprits include: global warming, the decrease in the mixing of the ocean waters, the oceans inability to pump the excess carbon from the surface to the deeper layers of the ocean, and the diminishing growth of the Phyto (plant) plankton (made to wander or drift) which are the foundation of the marine food webs22 are contributing to the increase in the ocean surface temperatures. This increase in the ocean temperature23 is negatively affecting the health of marine ecosystems. It is one of the major causes of coral bleaching. When corals undergo stress due to changes in temperature, light, and lack of nutrients they expel the symbiotic algae that inhabit their tissues. This process turns the beautifully colored corals white, hence the term coral bleaching24. The coral bleaching negatively affects the marine organisms such as fish that build their nurseries in these reefs, organisms that hide from their prey, decrease in revenue for tourism for some areas, and loss of diversity in the marine ecosystems. Figure-5 shows the journey of atmospheric CO2 from the atmosphere into the ocean waters causing ocean acidification, its impact on the shell formation of some marine animals, and how this process is devastating the entire marine ecosystem.
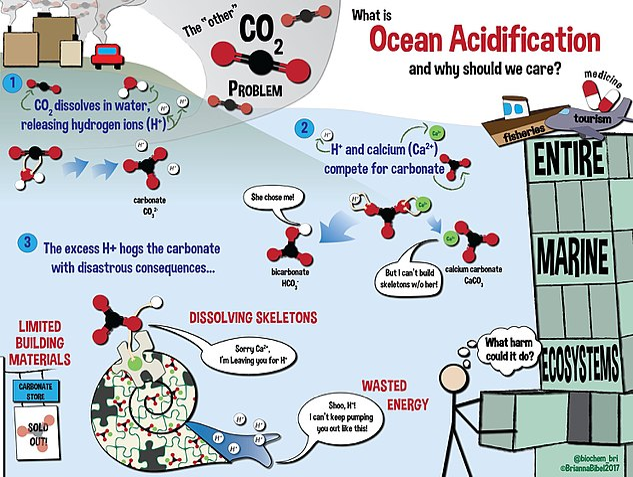
Figure 5: This infographic explains ocean acidification and its impact on the entire marine ecosystem. Adapted from Wikimedia Commons ( public domain)
Effect of ocean acidification on primary productivity: The process of ocean acidification negatively affects the entire marine ecosystem. For starters, algae and other photosynthetic organisms in the ocean undergo photosynthesis and store the organic carbon in their tissues. These organisms are the producers and form the bottom foundation of the food pyramid. The phytoplankton (free-floating plants)25 are consumed by zooplankton (pteropods) which are a major source of food for salmon, mackerel, herring, cod, and even whales. Organisms such as pteropods26 which are food sources for krill are having their shells dissolved due to ocean acidification. The krill are the main food source for whales. The consumers also undergo cellular respiration, use sugar to produce energy, and release carbon dioxide (CO2) into the ocean waters. When the phytoplankton dies, the carbon that is part of their tissues sinks to the bottom of the ocean. This process is called a biological pump. Therefore there is a relationship between the biological pump and the amount of ocean storage of CO2. Maintaining this pump is crucial to help decrease the amount of CO2 in the atmosphere. To summarize, the process of ocean acidification is greatly affecting the primary productivity which in turn, is causing a domino effect throughout the food chain, all the way up to the blue whales (keystone species), and threatening the existence of the entire marine ecosystem.
Effect of ocean acidification on soft shell formation of animals: The US Environmental Protection Agency (EPA)27 warns that changing ocean chemistry will 1) harm life forms that rely on calcium carbonate-based shells and skeletons, 2) harm organisms sensitive to acidity, and 3) harm organisms higher up the food chain that feed on these sensitive organisms. Ocean acidification is negatively affecting the number of carbonate ions in the ocean. Carbonate ions are used to form calcium carbonate, the main component of shellfish such as oysters, crabs, clams, and scallops. Lack of desired amounts of CaCO3 also affects the formation of eggshells, snail shells, seashells, pearls, and the shells of some soft-shelled animals that live in the oceans. Just as humans need carbonate ions to build their bones, several plants and animals that live in the oceans need to form calcium carbonate to build their shells and skeletons. Sometimes the shells are formed slowly and other times they get dissolved faster as compared to the time it takes for them to form. Thus the increasing acidity of the seawater has devastating effects on the survival, growth, and reproduction of these organisms28.
The ocean acidification process is often referred to as the osteoporosis of the sea. It is causing the shells and skeletons of marine organisms to become thinner, more brittle, and endangering the health and survival of these organisms. This process is also disrupting the marine food chain and damages could be traced throughout the marine ecosystem. Ocean acidification is also leaving these plants and animals vulnerable to predators and affecting their health and survival. The acidity is negatively affecting the survival of the larvae of several species. This process, if unchecked soon, could result in the extinction of marine plants and soft-shell-forming animals. Ocean acidification is negatively affecting fish, crabs, squid, oysters, and other marine animals too. The excess of carbonic acid is known to cause acidosis. This process is affecting the life processes such as respiration, growth, reproduction, development of larvae, echolocation, and the basic survival of these marine organisms. If this trend continues, then it could affect the predator-prey relationships in the oceans across the globe. It could also lead to the extinction of several species if they are unable to evolve and adapt to these new changes. Lastly, this could have devastating effects on the commercial fishing/seafood industries and the world economy.
Effect of ocean acidification on the coral reef ecosystem: Ocean acidification is one of the key threats to coral reefs29 as it reduces the calcification rate of reef framework builders. The acidity and decrease in carbonate ions are known to negatively affect the symbiotic relationships between the corals and the dinoflagellates that live in them. Scientists conducted experiments to see how ocean acidification affects coral reef-building organisms. The results from these studies revealed that the decrease in the pH of the oceans negatively affects coral survival, growth and calcification process, and their reproduction. It also causes the dinoflagellates to leave the coral resulting in coral bleaching30. Some scientists believe that coral bleaching could be reversed by decreasing the acidity of ocean waters.
It is promising to note that decreasing the amount of CO2 in the atmosphere could halt the process of ocean acidification and prevent coral bleaching. This demonstrates that we should as a collective should come up with strategies to decrease the carbon dioxide (CO2) released into the atmosphere. This could result in slowing down the global warming and ocean acidification process. The lesser the amounts of carbon dioxide (CO2) in the atmosphere the lower the absorption of CO2 by the oceans. This will in turn slow down the process of ocean acidification and in the process saving the plants, soft-shell-making organisms, the phytoplankton, coral reefs, and the entire marine ecosystems. This step could also help the biodiversity of our oceans, promote the fishing and seafood industry, local tourism, and prevent the mass extinction of marine organisms. According to the National Oceanic Atmospheric Administration (NOAA)31, ocean acidification, paired with the changes in the ocean temperatures are causing stresses on ocean life and the entire marine ecosystem. Ocean acidification has the potential to negatively impact the entire marine ecosystem and will certainly impact the fishing industry, tourism, and global economies. An overview of the marine ecosystem will try to explain how ocean acidification is affecting the producers as well as the consumers that are a part of this system.
The unit has the potential to inform the readers about the phenomena of the carbon cycle and ocean acidification. Understanding the concept of ocean acidification and how it is contributing to the decreasing of the pH in our oceans. Lastly, how this process is negatively affecting the plants, the shell-forming organisms, and the marine ecosystems. Some of the devastating effects include soft shell-forming organisms’ survival is threatened due to the weakening of their shells, affecting the primary productivity of the marine ecosystem, negatively effects the plants and animals that make up the marine ecosystem, loss of habitat, loss of diversity, coral bleaching, and extinction of marine life. The modeling of the carbon cycle, ocean acidification and its effect on fish and soft-shell forming organisms, and coral bleaching phenomena have the potential to not only help students understand these processes visually but also drive the concepts home. The students could be provided with opportunities to problem-solve and come up with strategies to mitigate, decrease, as well as reverse the harmful effects of ocean acidification. Students will have a chance to create awareness posters and share this information with friends. Lastly, allowing students to create awareness posters and write letters to their local representations about the harmful effects of ocean acidification in Connecticut32 will create enthusiasm about addressing the phenomena of ocean acidification. This unit could be modified for elementary and middle school level students.